DOCUMENT CONTROL
Document control that keeps pace with your team
Create, review, approve, and distribute controlled documents from one place. Version history is automatic. Every change is recorded in an immutable audit trail. Part of the Qualio compliance platform.

QMS document control software that does the heavy lifting for you
Challenge
Document management and control can be time-consuming and laborious - especially with a paper-based quality system.
From repetitive admin to chasing down signatures for approval, it can feel like you’re managing a daycare instead of building a real quality-centric culture.
Solution
Qualio provides integrated document lifecycle management software for your entire stack.
Standardize with templates.
Automate your QMS document control.
And embed real compliance with fully digitized lifecycle management and compliant
e-signatures.
Hybrid documentation control software to suit your business
- Make new QMS documents with our unique in-app editor, upload files in any format, or sync them straight from your company OneDrive
- Find the info you need in seconds with smart links and document tags
- Build bespoke workflows for document approval, training and review to drive the best document management system - without hours of admin and teams of personnel
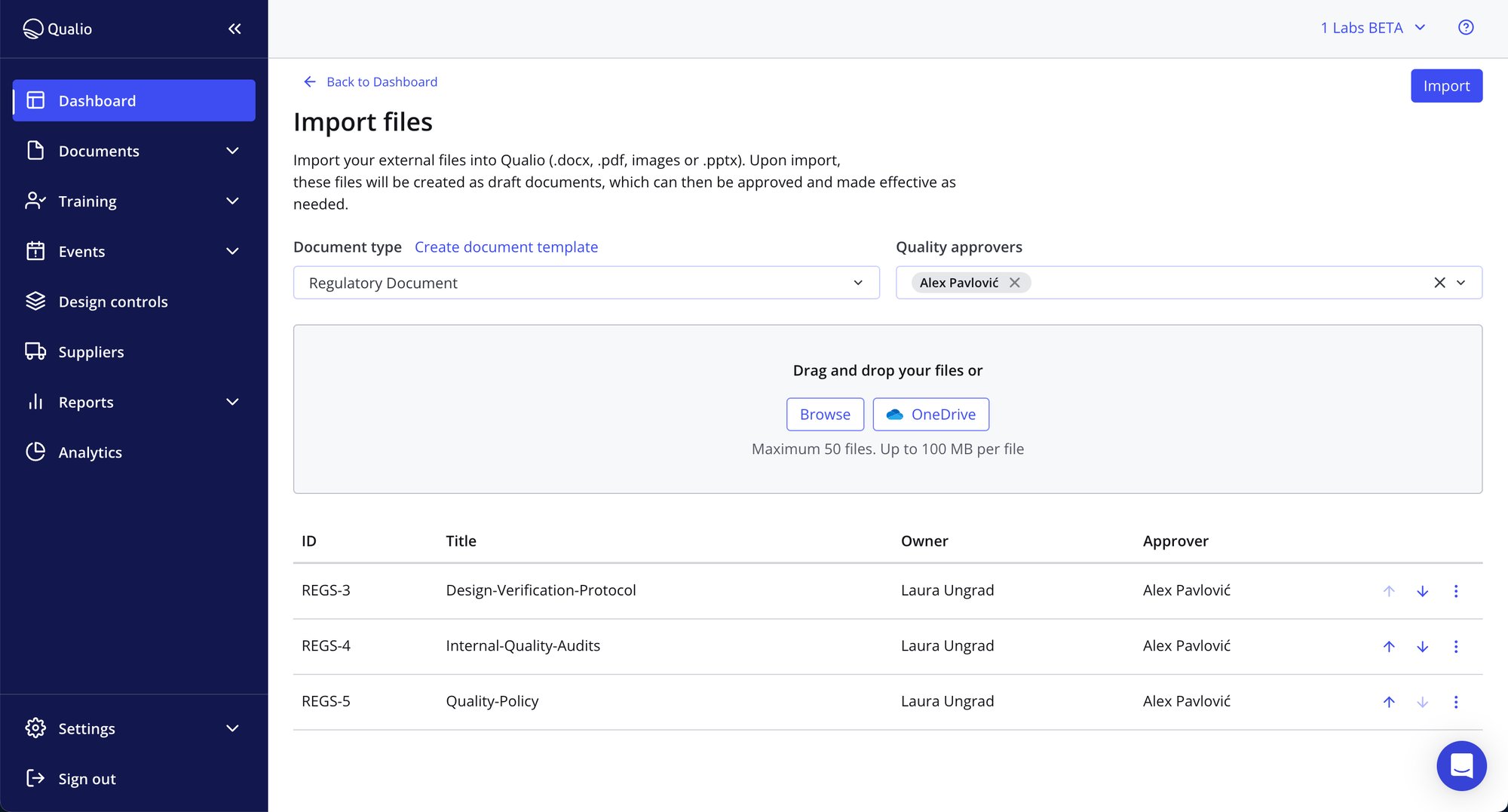
Data and collaboration in tandem
- Collaborate on document creation, distribution and review in real time with your colleagues - all from a single, centralized document management software system
- Secure your data by setting bespoke editing, view-only and download permissions
- Ensure your colleagues follow the processes you want them to with automatic version control
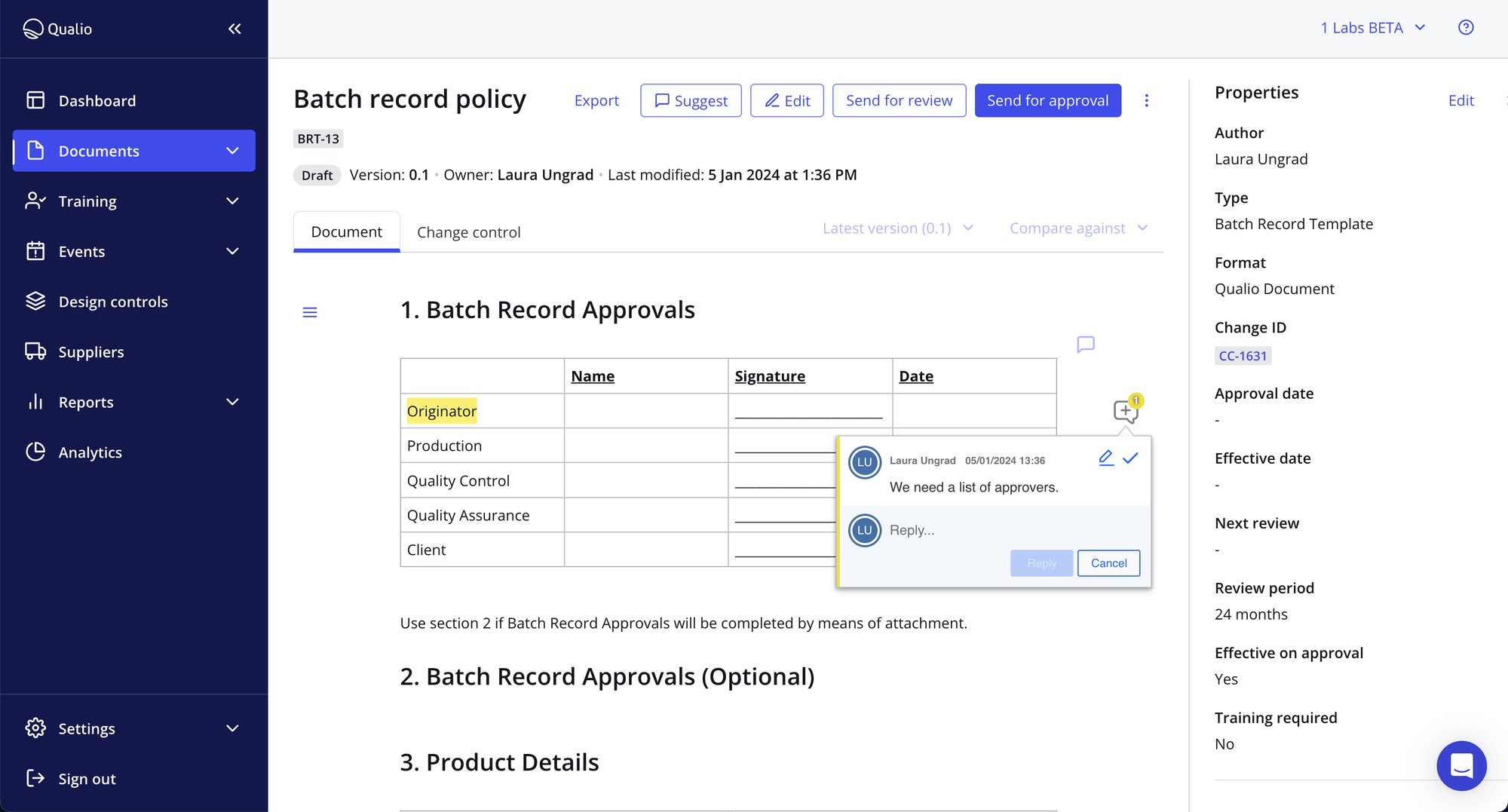
Airtight QMS document control with 100% compliance
- Meet your electronic record requirements with compliant and binding e-signatures
- Replace risky and insecure paper records with a fully controlled documentation control software platform
- Integrate with OneDrive to embed document compliance even as your colleagues work in other systems
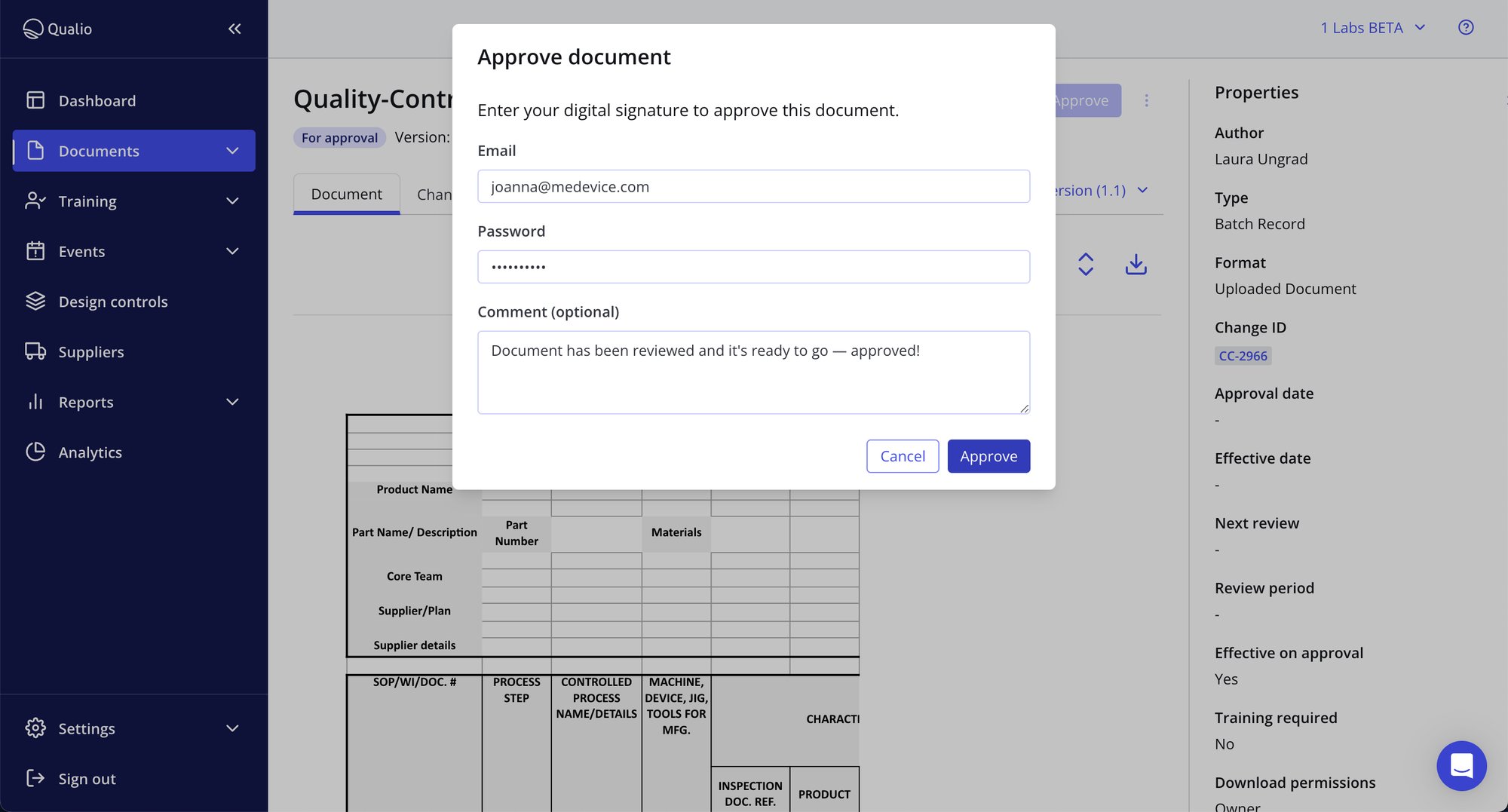
Build a QMS document management system with automatic best practice
- Meet ALCOA+ integrity principles with complete document lifecycle control
- Trace all document activity with incorruptible audit trails
- Demonstrate total compliance to your auditors with a purpose-built ISO document management software platform
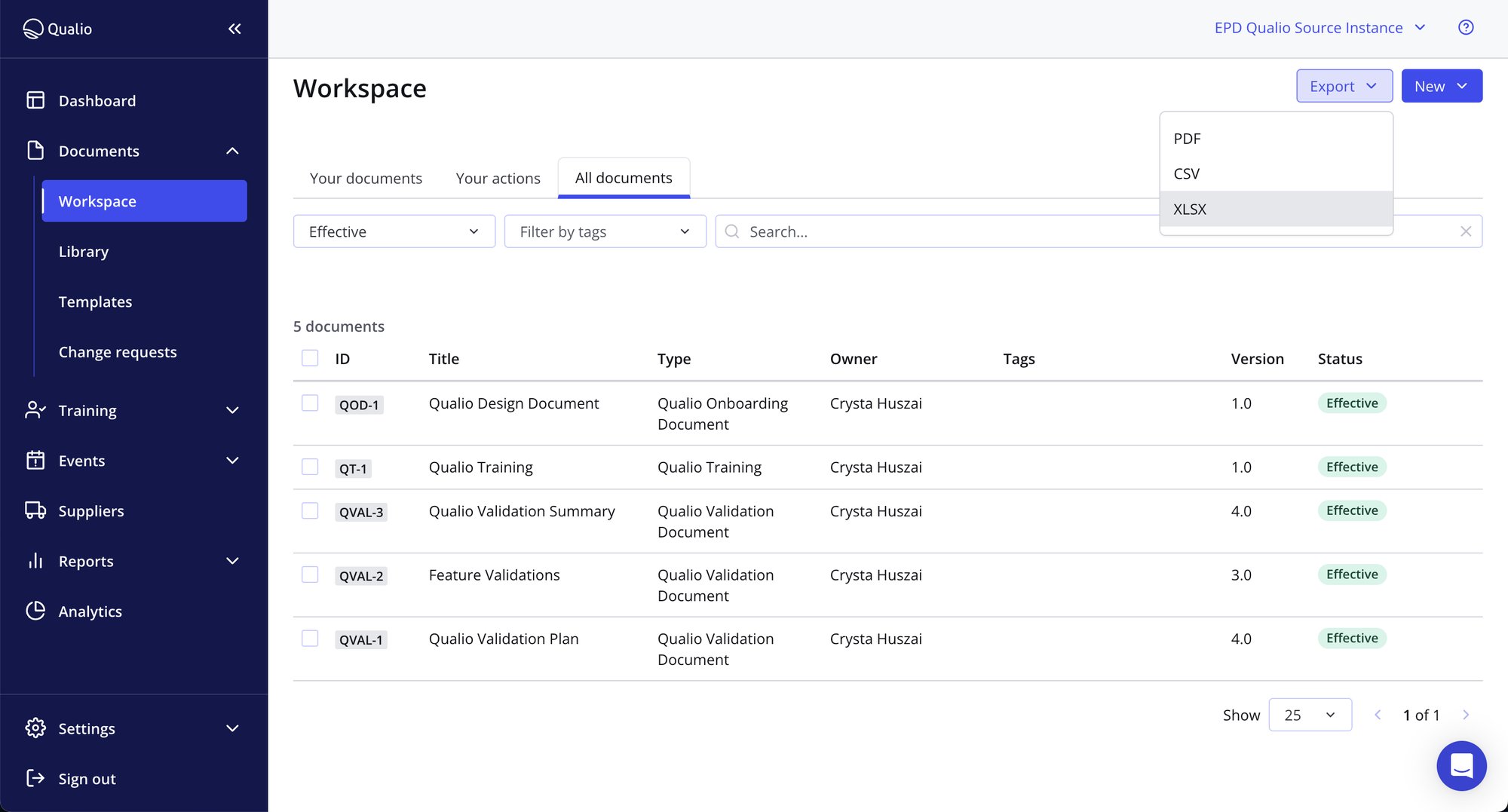
Document management resources

The case for document management
Read our whitepaper to learn the 6 key reasons your life science business should embrace software for document control.

Document management datasheet
Dive into the core functionality of Qualio's documentation management software and the operational benefits you can expect.
See the other features that make Qualio
the #1 top-rated QMS document management system
Design control management
Supercharge your product development from a single source of truth.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Change control management
Manage change activity easily and effectively with complete traceability.
Audit management
Make compliance and audit readiness the natural core of your business DNA.
Supplier management
Control and categorize suppliers and third parties with complete visibility.
The ultimate guide to document management software
Regulated companies with highly structured information flows are increasingly turning to document management software to make their lives easier.
It's little wonder. Document management software and document control software systems offer automation, traceability and control far beyond traditional methods like paper and spreadsheets.
And because document control is a critical component of modern quality management, it's crucial that regulated organizations, such as life science companies, get their document processes sharp in order to unlock optimal quality across the rest of their operations.
In fact, companies embracing QMS document control in tools like Qualio report more than a 90% reduction in their admin burden, as well as faster, easier audits and more time for value-add continuous improvement tasks. The knock-on effects of giving your business the best document management system can be dramatic.
Let's take a deep dive into document management software, and what it means for you.
Table of Contents
- Why is document management software so valuable?
- How to choose documentation control software
- What is document management software?
- What documents can you manage with QMS document control software?
- Document insights and analytics
- Do you need document management software?
- The return on investment of document control software
- The pros and cons of document management software
- Choosing the best document management software
Why is document management software so valuable?
Your employees lose at least half an hour every single day just searching for the information they need to do their jobs.
Connecting your employees to trusted, rapidly accessible information is one of the fastest ways to boost company productivity.
Searching cabinets, trawling through streams of emails and sifting through chaotic SharePoint folders waste employee time and energy, sapping general productivity and efficiency.
Weak document integrity, and multiple circulating versions, mean the wrong instructions are followed, processes are leaky and inconsistent, and accurately sharing trustworthy information becomes almost impossible.
Document management software eliminates these challenges.
And if you're in a regulated GxP environment, where good documentation practice and data integrity are non-negotiable, storing documents in a structured digital environment is the perfect way to set guardrails and guarantee constant, long-term compliance.
It doesn't hurt that a clear, digitized document management quality system also proves to your external auditors that you've got things under control, and cuts audit timeframes fivefold.
How to choose documentation control software
How do you choose the best document management software for your specific business needs?
Having a set of standard questions to ask yourself and potential vendors is the best way to start the process.
It's also important to note that document control software is often bundled into a broader set of functionality, such as a quality management system software platform.
To minimize the number of separate, siloed systems your company uses, it's usually worth investing in one of these broader, more holistic systems that offer QMS document control alongside other important compliance features like training management and CAPA and nonconformance management.
Qualio's 12 questions to ask before you buy an eQMS is a good way to pinpoint your company's exact documentation management software requirements, then gauge the ability of each eQMS vendor to satisfy them:
What do you want to achieve?
Does it meet your industry needs?
Does it meet your organization’s size and scaling needs?
How customizable and flexible is it?
What 's your budget?
How long will it take to get up and running?
Will it support your compliance needs?
Are onboarding services included?
What’s the support like?
Will your important data be secure?
Will it help you go paperless?
Is it easy to use?
What is document management software?
Document management software and document control software are usually used interchangeably as terms.
In short, software for document control is any digital system that offers dedicated functionality for managing the so-called 'document lifecycle'.
The document lifecycle of creation, collaboration, distribution and archiving of a document is an important cornerstone of modern quality and compliance, because documents offer a standardized blueprint of how your company works - and evidence that you're doing what you're supposed to when regulators check up on you.
Document management software should offer features like:
-
Digital and online 'paperless' document storage, often in the cloud for remote access
-
Collaboration features, like inline commenting and mentioning to support input from multiple owners
-
Version control, e-signatures and audit trails for compliance with electronic record regulations like GDocP or FDA 21 CFR Part 11
-
Permission control, so only certain users can view a particular document
-
In-app editing and updating, to reduce reliance on 'checking into' and 'checking out' of non-compliant systems like Word
-
Integration with other tools like OneDrive for easy upload and syncing of changes
The best document management system for your business should offer all these features. If not, perhaps it isn't delivering the best value for you!
What documents can you manage with QMS document control software?
Documentation control software should empower your business with the freedom and flexibility to manage any kind of document you need to.
QMS document control and ISO document management software is typically deployed for storing and distributing critical quality and compliance documents, such as:
-
Policies
-
Standard operating procedures (SOPs)
-
Work instructions
-
Organizational charts
-
Process diagrams
-
Audit records
-
Equipment and calibration records
-
Medical device design documents
And more.
The best document management system software, like Qualio, should make it as easy as possible for your company to work with standardized, controlled versions of these key documents without hassle or fuss.
That's why Qualio offers pre-built document templates and content pathways to accelerate your compliance and set repeatable, audit-ready document frameworks for you to start using straight away. Accessing these ready-to-populate document templates eliminates around 6 months of document building work, freeing up our customers to work on other things.
Document insights and analytics
Another key benefit of documentation management software is its ability to provide bird's-eye visibility of how your documents are really being built and used day-to-day.
It's impossible to access analytics and digital insights for a piece of paper - but digital documents, living in document control software, can be both controlled and monitored to give you the best document management quality system possible.
Document platforms unlock powerful compliance analytics such as:
-
Who has read, been trained on and signed off on a document
- Document cycle times and approval timeframes
- Statuses of documents across your business: live, in draft, archived, and so on
- Due, overdue and completed document training
- Open actions and bottlenecks with responsible personnel
Document management software can therefore become a tool for continuous improvement and business-wide accountability - not just a digital version of a filing cabinet.
Do you need document management software?
That depends.
If you've ever struggled with any of these pain points, document lifecycle management software could be right for you:
-
Employees struggling to find information (or losing it)
- Duplication of work and unnecessary copying of documents
- Awkward filing cabinet retrieval trips while an impatient auditor waits
- Physically large/confusing paper document storage areas with manual upkeep requirements
- Long email chains with back-and-forth attachments and uncontrolled document versions
- Suboptimal GDocP, ALCOA+ or Part 11/Annex 11 compliance
- Time-consuming wet signature-gathering
- Siloed paper, Dropbox, email and SharePoint systems
You could try and work around these problems by doubling down on your legacy system. More filing cabinets, more document personnel, going paperless with a free system like SharePoint.
Or you could invest in documentation management software and save money in the long run.
How? Read on!
The return on investment of document control software
Investing in the right QMS document control can deliver transformational ROI.
Consider this:
The average company spends $20 on labor to file one document.
Another $120 locating a misfiled document.
And $220 replacing a lost document.
When you take into account industry estimates that about 7.5% of paper documents get lost, and 3% misfiled, these labor and time costs can slowly mount up to considerable expenditure and wastage.
There's more. The average annual cost of filling and maintaining a single filing cabinet? $2,000.
The annual production, usage and storage costs of hard documents - just for a small business? That's around $25,000.
And for larger businesses, it's more like $175,000.
Document management software eliminates or dramatically reduces all of these costs, freeing your company of manual upkeep work and physical storage while accelerating document processes to maximum efficiency.
In fact, one Qualio customer estimates that her documentation management software does the work of at least 5 full time employees (FTEs). This represents a six-figure headcount cost saving to be applied elsewhere.
Another saved around $30,000 in consultancy costs by accessing Qualio's pre-built document templates and targeted expert guidance.
Users of Qualio's document management application report:
-
>90% reduction in document admin and location time
-
25% faster document lifecycle processes
-
Up to 5 FTE spends mitigated
-
5x faster external audits
-
Simplified compliance with FDA 21 CFR Part 11, GDocP, ISO 27001, ALCOA+, and other document integrity standards
- More collaborative quality cultures built on controlled information
The pros and cons of document management software
Before you act, it's worth weighing up whether documentation management software is the right choice for your business.
Like any tool, your QMS document control system needs to be carefully evaluated and compared with your existing set-up before purchase. To help you, we've assembled a handy list of the pros and cons of documentation control software.
Pros of document management software
There are plenty of benefits to installing document management software. Let's start with those.
Stronger compliance
Document control software gives you exactly that: control. Whether it's ISO 27001, GDocP or FDA 21 CFR Part 11 compliance you're aiming for, it's much more difficult to achieve it with manual legacy document systems.
The best document management software makes paperless compliance a natural and automatic part of your day-to-day, with features like e-signatures, version control and audit trails forcing employees to act in a way that maximizes your data integrity and traceability - with only the latest and greatest versions of documents, such as SOPs, available for viewing.
More time for value-add
We discussed above how much time gets wasted just looking for documents in a manual or paper-based system.
Half an hour per day searching for information is the equivalent of 1 in 16 of your employees doing no productive work at all!
Software for document management provides cloud-based paperless document access at your fingertips, with features like categorization, smart linking and tagging helping to build a connected web of information that makes employees more assured, confident and productive.
Cultural change
Closely connected to this newfound accuracy and control of information is the shift to a culture of quality and compliance without employees even realizing it.
Any documentation management software you use must be intuitive and require minimal user training. With strong internal messaging, this will allow you to drive internal operational changes that are as much cultural as procedural:
-
If the document is not in the document management system, then the work wasn’t done
- Paper documents are uncontrolled and should be minimized and phased out
-
Always use documents or forms directly from the system, not copies that you downloaded months earlier
-
Bookmark your document management software system and take 10 seconds to check it each day for any outstanding document reference or training you need to complete
Integration
Paper, emails and Dropbox documents just sit there, disconnected from your broader business processes and squirrelled away in drawers and behind private account logins.
At the same time, siloes naturally begin to form between your quality teams and other key departments like engineering and product development.
Any truly transformational document lifecycle management software should smoothly integrate with as many of these existing systems as possible to allow instant business-wide access to key information without the need for extra searching or duplicated storage.
Functionality like Single Sign-On breaks down the barriers to a single source of truth.
Document integration functionality allows documents generated in typical tools like SharePoint to be absorbed into the system quickly and easily.
Plus: the best document management systems like Qualio also offer powerful integration functionality with tools like Asana, Jira and Azure DevOps, allowing key documented information like design controls, product requirements, test cases and trace matrices to be pulled into a centralized information repository.
Gold dust when your auditor or a key potential customer comes knocking.
Cons of document management software
Why might you be better off without document management software?
Upfront cost
Of course, document control software isn't free. Typically, it's paid for with a monthly or annualized software-as-a-service (SaaS) subscription to your vendor of choice.
Basic tools cost a few thousand dollars a year, while more comprehensive platforms with integrated quality management functionality for regulated companies represent a five-figure investment.
Of course, paper-based and manual document control systems have a slew of hidden costs - but they are exactly that. Hidden.
It can therefore be difficult to convince your procurement team of the benefit of paying for a system to replace something that's theoretically 'free' or low-cost.
To do that, you should lean on the powerful return on investment capabilities of a document management software system that we've already discussed.
Training
It's likely that every single one of your employees knows how to build a Word document, print it, store it, or save it in a repository like SharePoint.
Document management software can be different. Depending on its scope, complexity and design, any investment will also require at least some training before it can be rolled out and deployed.
To sidestep this issue, look for document control software specially designed for ease of use and rapid implementation - as Qualio's G2 accolades show. The more intuitive and user-friendly your software is, the quicker you can start seeing benefits - and the more of your team will consistently adopt it.
Configuration & validation
In a similar vein, your documentation control software system will require some configuration and validation before going live in a regulated GxP environment.
Like with training, your requirements will vary. GAMP Category 5 software systems with high levels of configuration, customization and 'bells and whistles' can be powerful and adaptable, but can require months of adaptation and validation work.
GAMP Category 3 systems are rigid and inflexible, making them perfect for rapid roll-out with minimal configuration and lighter validation. This rigidity, though, makes it more likely that you'll outgrow the system as your company and processes evolve - forcing you to start looking again in a year or two.
The sweet spot for your company could therefore be a GAMP Category 4 system: scalable and configurable enough to match your evolving company, but without the complex, time-consuming malleability and endless validation demands of a larger platform.
Going paperless
Hang on, I thought you said going paperless is a good thing!
Of course it is - but making the transition from paper to eQMS isn't necessarily easy with every document management software vendor.
If you have rows of filing cabinets, stacks of paper, and even folders of digital documents, porting them across to your shiny new document control software platform can be a tricky task.
It's therefore crucial to quiz vendors on their migration and support capabilities. Most vendors will help you identify and bring over the legacy documents you want to keep, but the depth and quality of support will vary.
It's also a good idea to look for a document management software system that makes migration, management and upload of your legacy documents as simple as possible.
Qualio, for instance, pairs high-touch, award-winning migration support with hybrid document management functionality that allows you to:
- Sync documents straight from OneDrive - your teams get to keep accessing documents in familiar places, with any updates automatically synced into your eQMS for compliant review and approval
- Bulk upload documents in their original formats, from diagrams to SOPs
- Convert uploaded documents into fully templated and controlled system-format documents linked to the originals
Flexibility can be key to your document management software and your paperless journey.
Choosing the best document management software
Now you've read this far, it's time to start the exciting work of scoping out the best document management software for your company's needs.
Start by working through our 12 questions to ask before you buy an eQMS.
Pinpoint the features you need, the kind of vendor you want to work with, and whether you need a system purpose-built for document compliance and data integrity.
Identify a shortlist of 3-5 optimal vendors, then check out their G2 reviews to see if they're really doing what they say they are for their customers.
Then it's time to start the best part: booking in for a live demonstration of each platform.
Start your document management software journey by booking in for your Qualio demo below 👇