FDA COMPLIANCE SOFTWARE
FDA compliance software for long-term US market success
Make your FDA inspector happy.
Get your drug or device cleared first time.

FDA compliance software for constant audit readiness
Challenge
The Food & Drug Administration is the life science gatekeeper for 330 million people in the world's largest economy.
Getting your life-saving product to market means hawk-like inspection in accordance with some of the world's highest quality and compliance standards.
Some companies never make the cut.
Solution
The FDA wants life science companies to digitize their quality approach.
Qualio makes FDA compliance natural and automatic by surfacing gaps, sharpening visibility and unlocking complete control of your quality and compliance processes.
Electronic quality =
unshakeable FDA compliance.
Used by 700+ companies for compliance with...

FDA compliance software: how it works
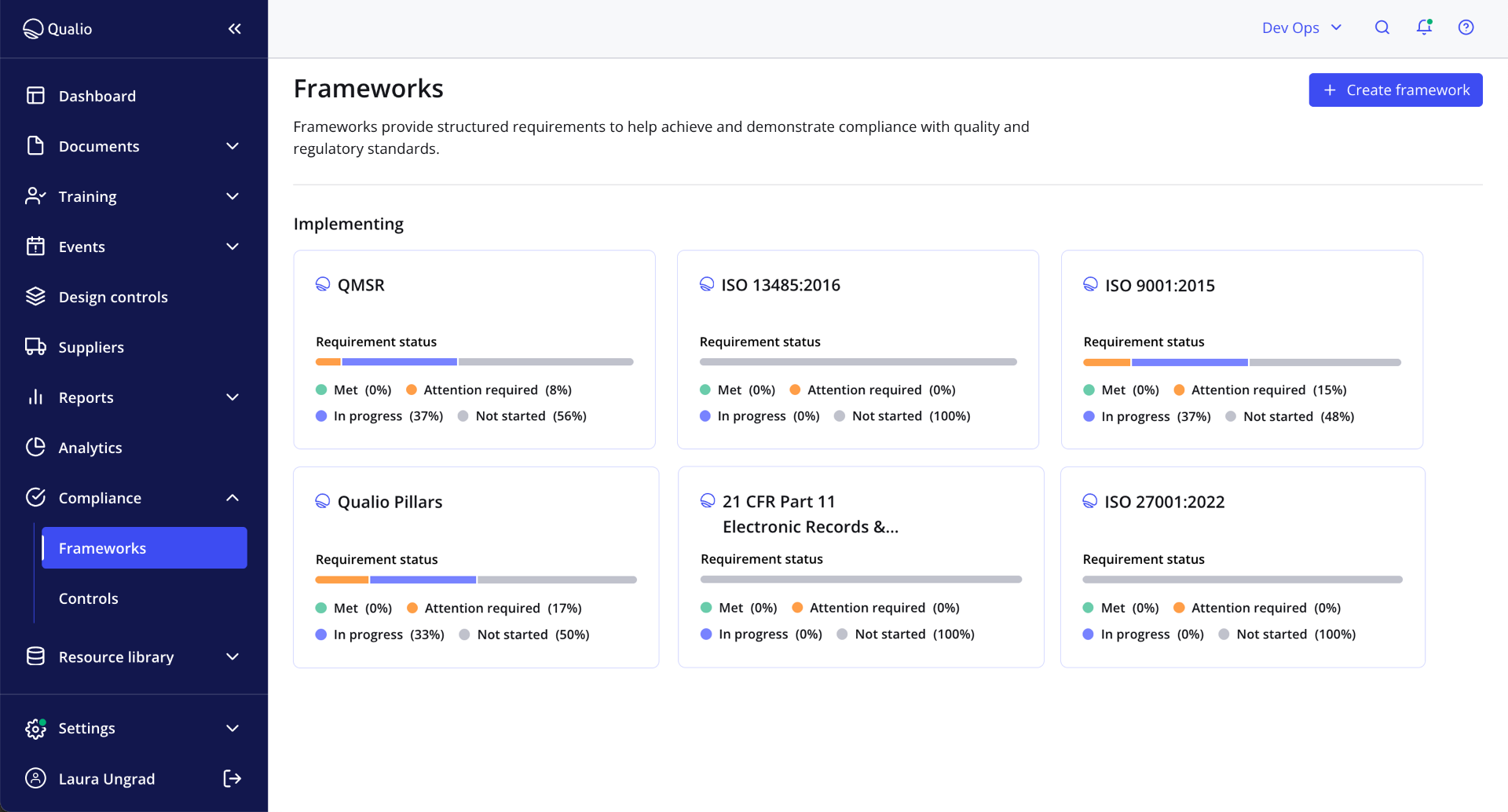
Automatic gap analysis for total audit readiness
- Automated gap analysis scans your entire quality system in 30-40 minutes, highlighting every FDA compliance gap in your organization — from the QMSR to Part 211 cGMP
- Allocate tasks and automatically gather evidence to get 100% audit-ready in weeks, not months
- Enjoy full FDA inspection confidence with constant monitoring and compliance health dashboards
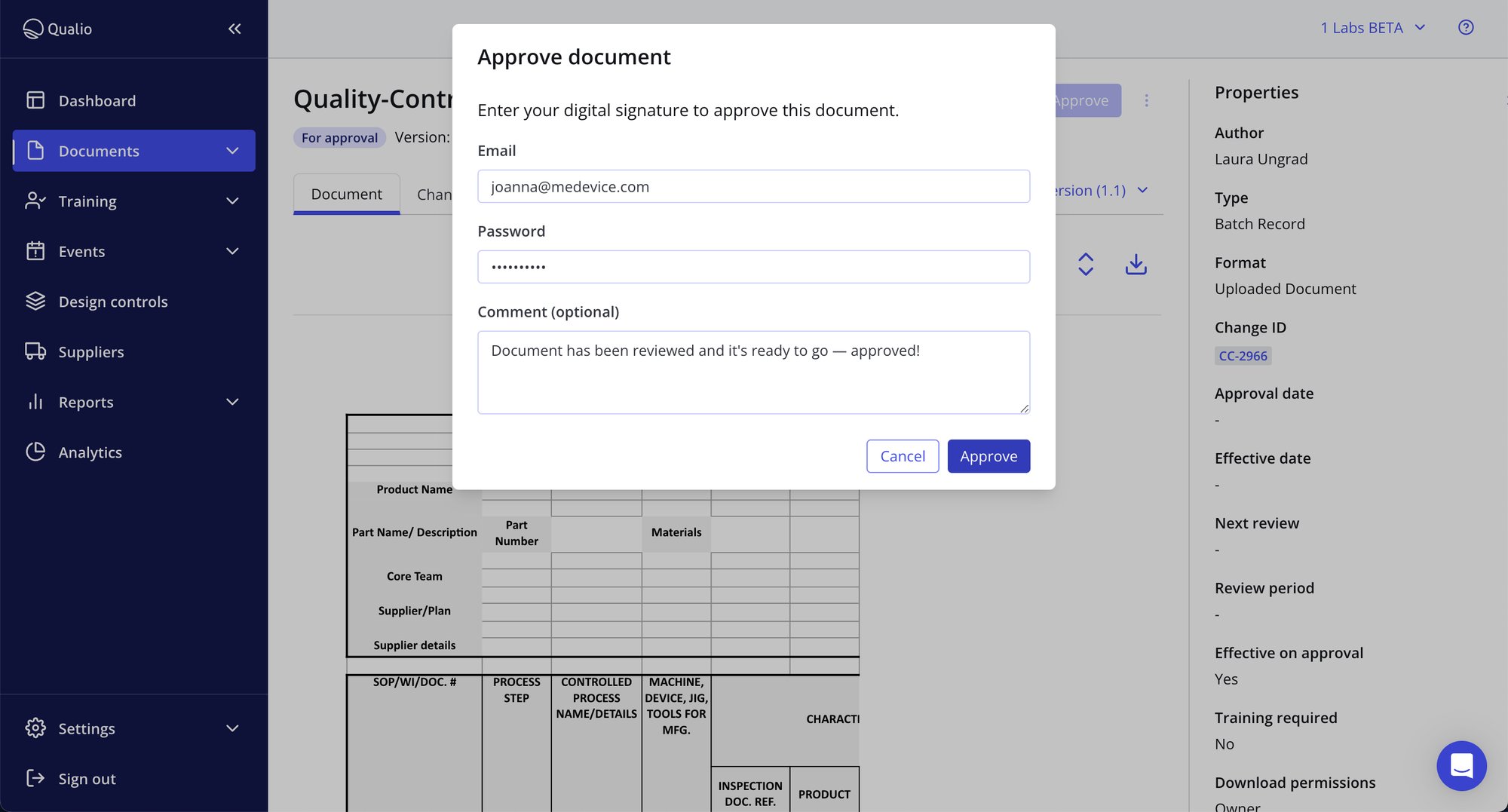
FDA compliance baked in
- All major system actions, from document review to training completion and event close-out, underpinned by Part 11-compliant e-signatures
- Incorruptible audit trails give complete traceability and accountability for your inspector
- GDocP and ALCOA+ system guardrails give you a controlled, compliant single source of truth for all your quality records
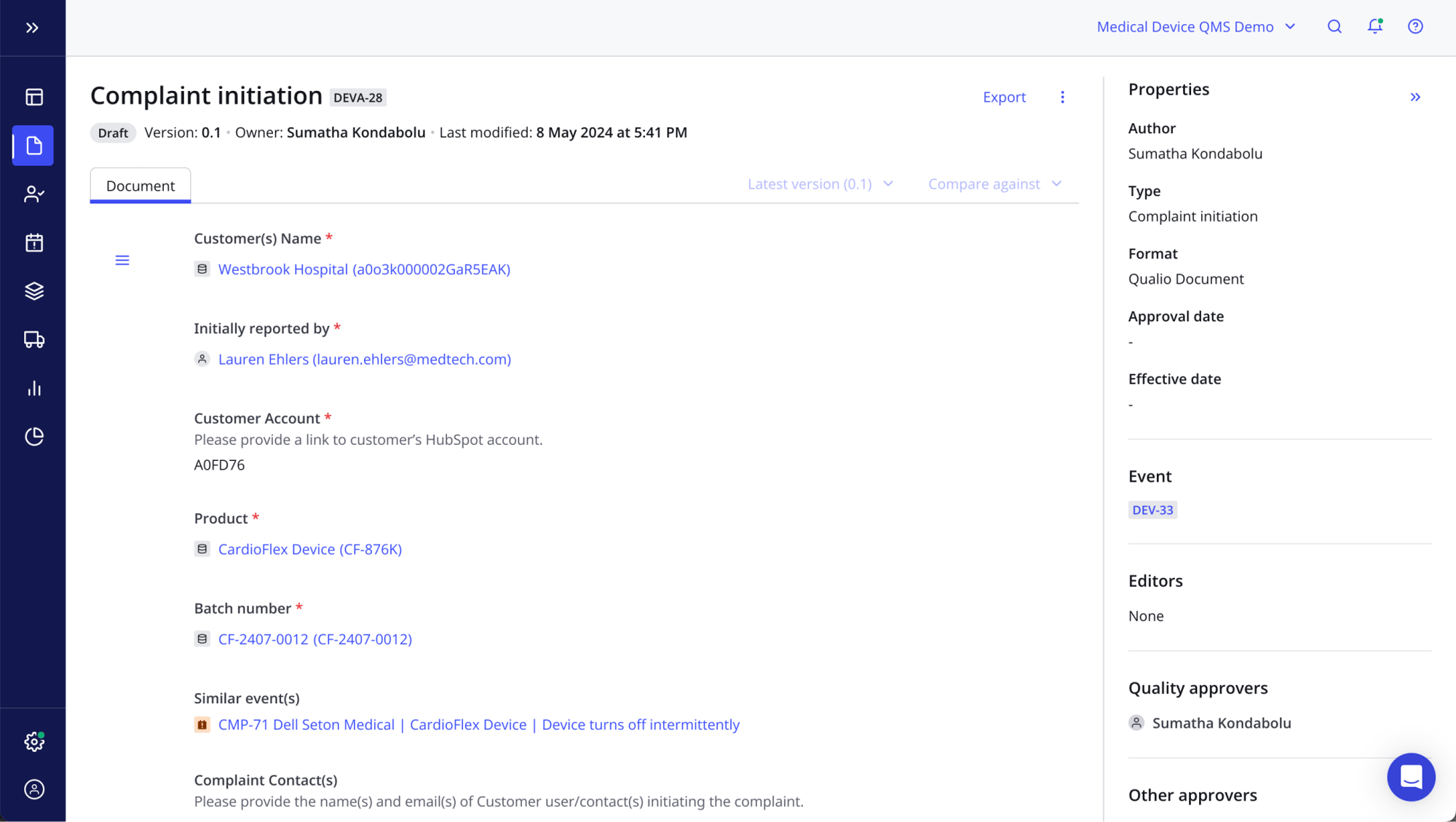
Integrated eQMS
- Access all the elements of a modern, FDA-compliant quality system, from document control to training and CAPAs, from a single platform
- Feed pre-built content and templates into your eQMS to accelerate your maturity
- Easily combine and control all the ingredients of your 510(k) or PMA submission, from device safety data to substantial equivalence documents
FDA clearance resources
Everything you need to know about 510(k) submissions
Access our comprehensive end-to-end guide for your medical device's FDA 510(k) submission.

What your QMS needs for
FDA & ISO regulation
Do you know what your QMS needs to make your FDA inspector happy? Find out with our webinar.