ISO COMPLIANCE SOFTWARE
ISO compliance software for long-term international success
Get ISO certified. Unlock markets. Embed lasting compliance.

ISO management software for easy compliance
Challenge
ISO standards provide the globally recognised quality benchmarks for regulated companies.
Businesses focus the entire energy of their quality systems towards compliance, leaving no time for real quality improvement and turning audits into stressful nightmares.
And with quality contained in paper and spreadsheets, a fatal lapse in compliance remains a constant risk.
Solution
Qualio's ISO compliance software makes meeting ISO requirements natural, automatic and easy.
Automatic, AI-powered gap analysis and remediation tasks, all in a centralized and connected eQMS, ensure every ISO compliance gap in your organization is found and fixed.
Audits become routine, and ISO compliance becomes unshakeable.
Used by 700+ life science companies for compliance with...

ISO compliance software for multi-standard confidence: how it works
Automatic gap analysis for total audit readiness
- Automated gap analysis scans your entire quality system in 30-40 minutes, highlighting every ISO compliance gap in your organization
- Allocate tasks and automatically gather evidence to get 100% audit-ready in weeks, not months
- Enjoy full ISO confidence with constant monitoring and compliance health dashboards
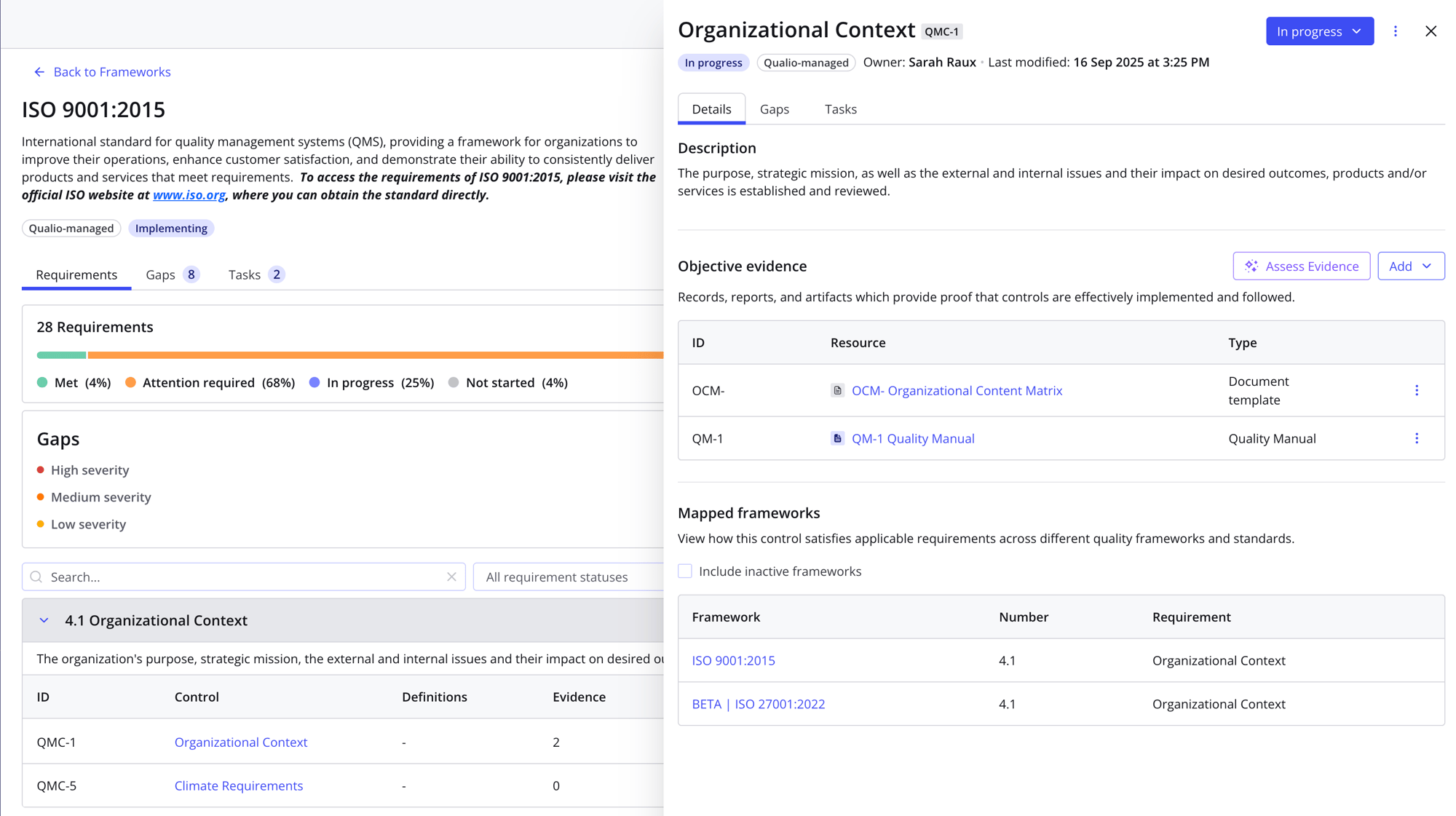
Integrated eQMS
- Connect all the elements of a holistic ISO-compliant QMS, from document control to training, in a single cloud-based system
- Pull product design and testing data straight from their source systems with instant integrations
- Build flexible workflows for approving documents, checking suppliers, managing CAPAs, and more

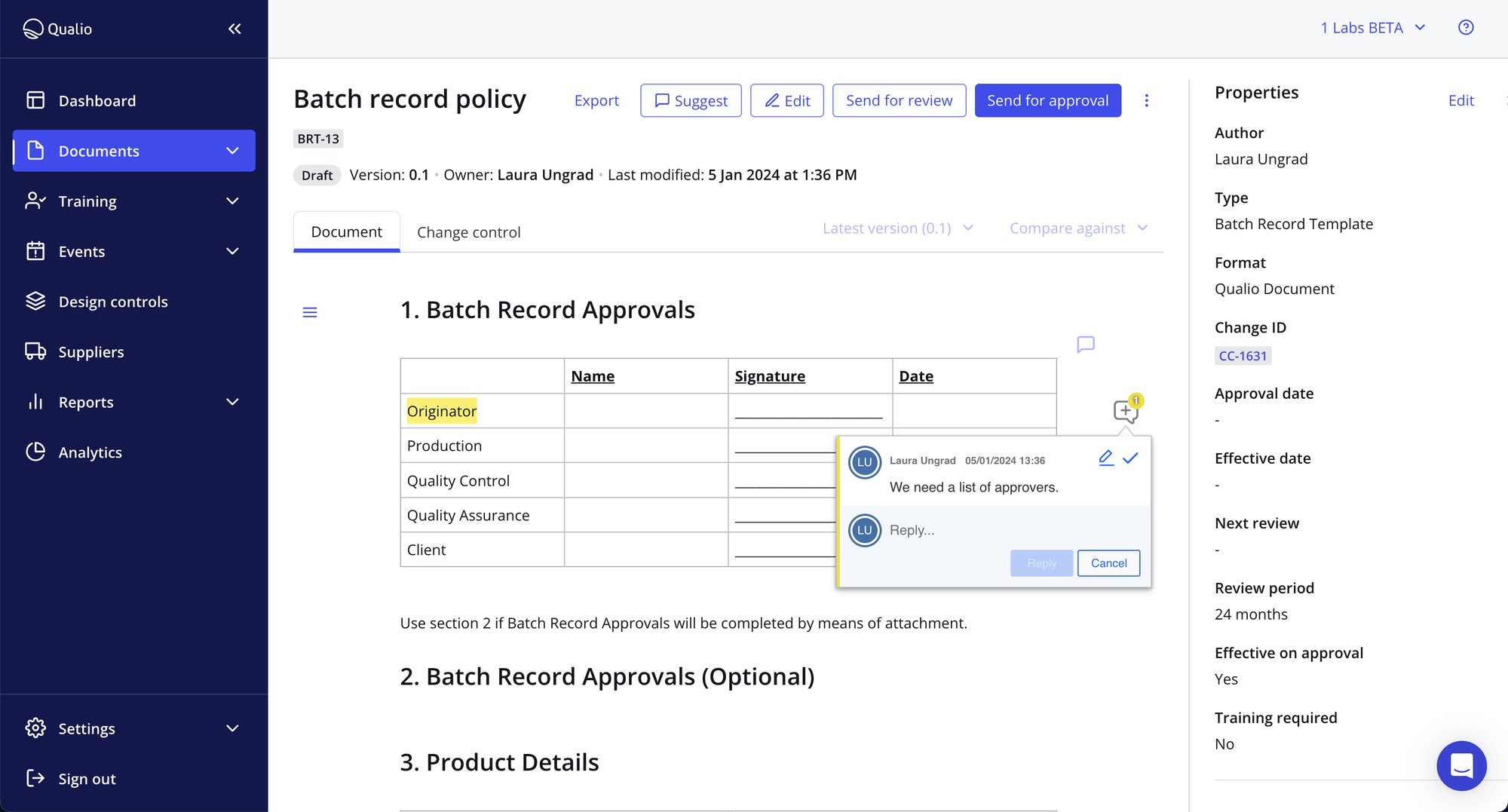
Business-wide compliance
- Single source of truth aligns your business around key ISO requirements, from proving management commitment to managing resources and instilling customer focus
- Collaborate on quality & compliance tasks with ease, from building documents to signing off on quality events
- Audit trails and connected records prove your ISO compliance to customers, regulators and auditors
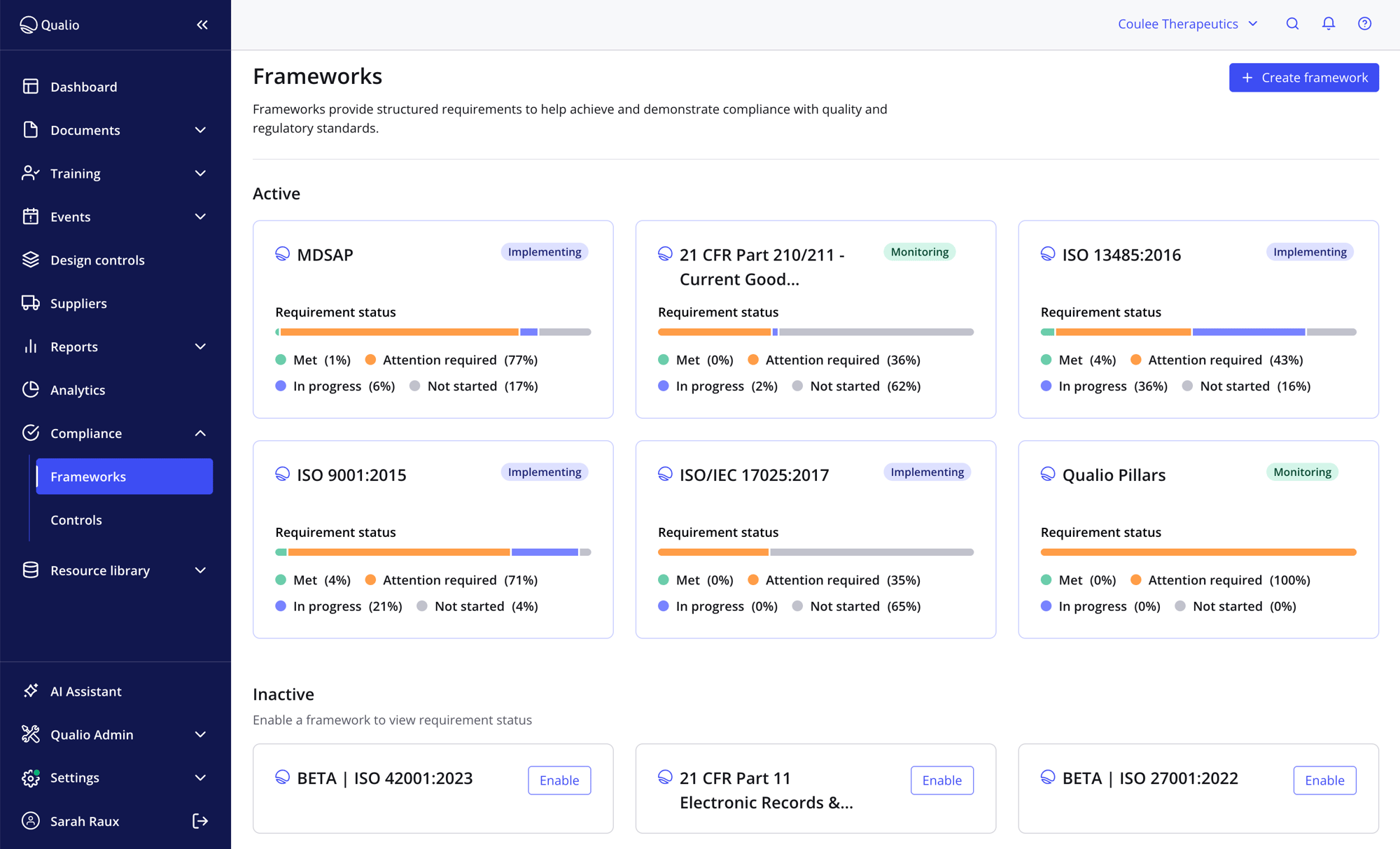
Meet the standards that matter to you
- Cut consultant spend with pre-validated, expert-built ISO frameworks
- Reuse evidence across multiple standards, audits and markets to slice compliance burden
- Track compliance progress for your ISO standards at a glance
ISO management software resources

ISO 13485 toolkit
Learn how to tackle the medical device quality management standard with our bundle of helpful resources

ISO 9001 toolkit
Become an expert on the core quality management standard: the baseline for all other ISO standards.