The Agentic Compliance Platform for Life Sciences
AI-powered compliance for life sciences, medical devices, SaMD, GxP Software, pharma, and biotech. AI-powered compliance for medical devices life sciences
Accelerate product development with an AI-powered quality and compliance platform. AI agents run gap analyses, monitor regulatory changes, and execute workflows to stay audit-ready and bring products to market faster.
Request a demo
FROM 20+ WEEKS TO <4 FOR AUDIT READINESS
Compliance Intelligence
Automate gap analysis across FDA, ISO and EMA standards, along with 28 others. Cross-map evidence once, reuse across submissions. Get real-time risk alerts and board-ready dashboards, so you can expand into new markets with confidence.
Proven Results: 80% reduction in audit prep time | 5x ROI in 2 months | 60% lower consultant spend

THE QUALIO PLATFORM
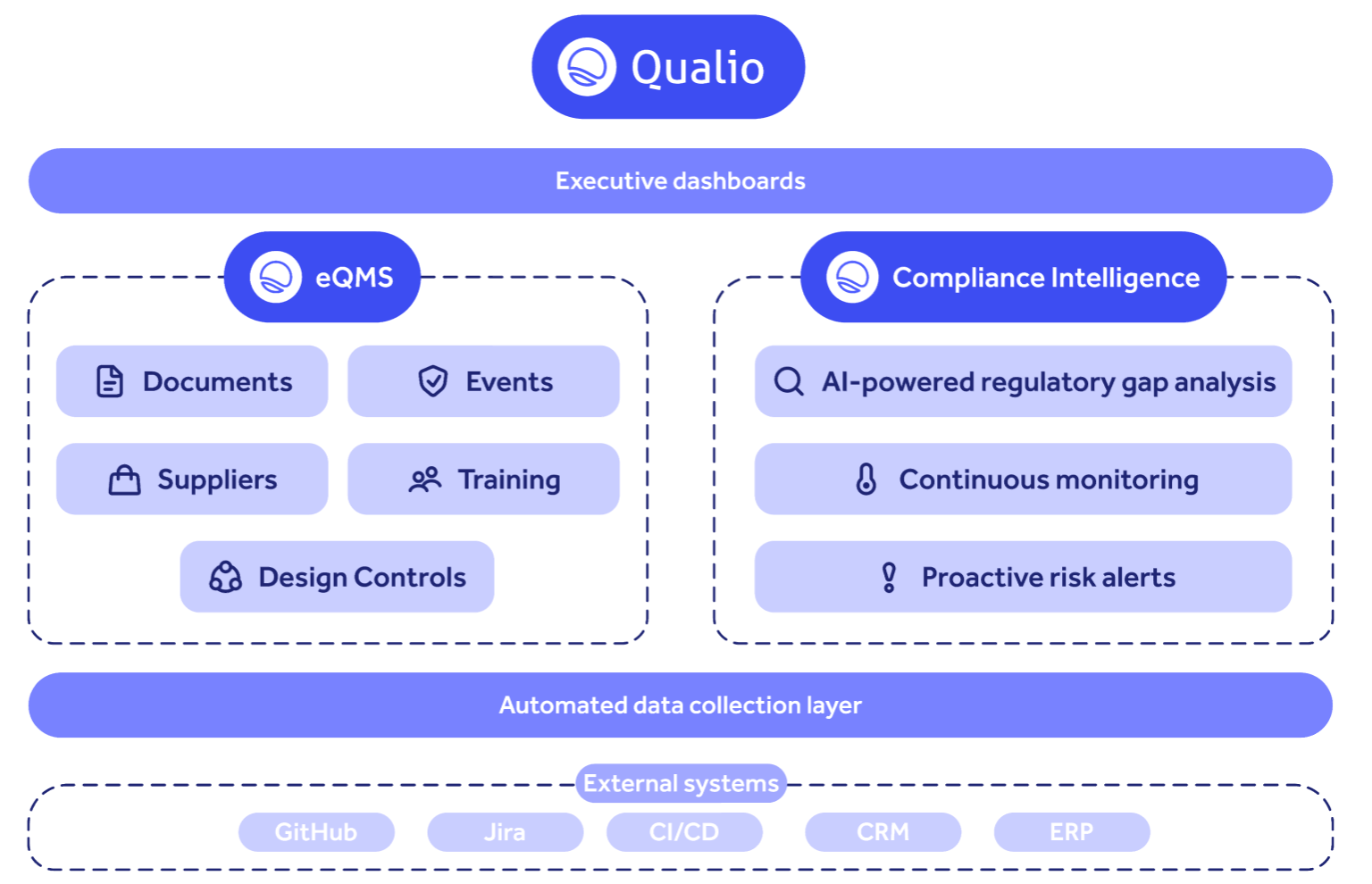
How the modern compliance platform works
The bar has never been higher for life science companies.
Every fresh regulation, new product you launch or new market you target adds to a complex web of regulatory demands.
Qualio unites quality management, product lifecycle and compliance intelligence in a single, integrated platform. With AI-powered gap analysis, cross-mapped evidence and continuous monitoring, we get you audit-ready in weeks instead of months — so you can launch faster, protect revenue and scale globally with confidence.
Trusted by 700+ highly regulated customers across the globe

SMARTER COMPLIANCE
Constant audit readiness in a world of changing regulations
Our customers use our software to comply with:

QUALIO SOFTWARE
Purpose-built for life science companies
Qualio serves the entire life science ecosystem.
Find out how we support your specific industry.
Medical Devices
Streamline a fast, safe pathway to market for your medical device while ensuring your teams stay compliant.
Pharmaceutical
Embed industry-leading pharmaceutical quality processes that simplify and unlock FDA, ISO and ICH compliance.
Biotech
Beat the competition with quality-centric products that satisfy regulators, patients and stakeholders.
Cannabis
Earn regulatory trust and position your business for growth with a robust and quality-first approach.
Software as Medical Device
Unlock end-to-end control of your entire product lifecycle, connecting teams and cutting stress.
Contract Research Orgs
Deliver a compliant and continuously improving CRO service that delights clients and brings new ones flocking.
Our validation approach
- We do the heavy lifting, so you go live 2 weeks faster than traditional eQMS validation methods
- Built around the latest FDA and GAMP industry guidelines
- Pre-built templates and automated document packs accelerate or eliminate your validation tasks
- Only 1% of our customers choose to validate themselves!

Don't just take our word for it!
Hear from our many happy customers
"Qualio is the best QMS cloud-based system I have ever seen."
Paul H.
Senior VP of Ops, Tech & Quality, Advantice Health
"You're going to be able to get this thing in-house, validated, implemented and live in a short amount of time with limited resources.
That's unique in this industry."
Becki N.
Quality & Regulatory Lead, Synthego
"We were able to complete a demanding audit thanks to Qualio. The smart links made navigating between processes seamless and easy to understand."
Thais S.
Head of Quality & Regulatory Affairs, Cergenx
“Qualio has helped make my life as a quality manager very easy.”
Gene V.
Quality Manager, Cirris
"We looked a lot at how Qualio worked, its user interface, and the technology behind it. It was by far the freshest, most logical eQMS system out there."
Dmitry F.
Chief Technology Officer, ViQi
"I contacted Qualio in November. We went live mid-December. We had all users in by January. And we had our ISO 13485 certification in July.
Without Qualio, we'd never have done that."
Karen H.
Head of Quality & GxP Compliance, 30 Technology
"Qualio’s become a verb here. When people need things, Qualio is the word that’s used."
Chris R.
QA Associate, AGADA Biosciences
"There are other eQMS providers that are very well-established, but not particularly accessible.
Qualio is not overly complicated. It gives us those foundational aspects of a quality system in a way that people can actually get behind."
Phil C.
Quality Manager, Bond Digital Health
"The perfect QMS system for a start-up. Everything, from validation to migration and training, was a positive experience."
Drew M.
Director of Quality, ECM Therapeutics
"Me getting back hundreds of hours in a year thanks to Qualio is the equivalent of an FTE.
Qualio has given us one more staff member in a company where we throw nickels around like manhole covers!"
Mike T.
VP of IT & Information Security, xCures
“The cost of our Qualio licenses is insignificant compared to what we've saved by just improving our processes.”
Peter B.
Director of QA, Watchmaker Genomics
"The Qualio team made a big difference to our company by providing amazing help and guidance at a critical time in our QMS development."
Jackie L.
Managing Director, Auxilio Pharmaceuticals
"I entered a world with Qualio where quality is no longer a hassle."
Fedora B.
Co-Head of Biopharma Corporate Finance, JMP Securities
“We evaluated all players on the market and came up with Qualio as the best balance of price, capability, and quality of support.”
Jeffrey B.
CEO, FifthEye
“Once you understand how Qualio works, it's really easy to plan out processes and put them into place within the QMS.”
Sara V.
Quality Manager, Leaseir Technologies
"Qualio keeps us in a constant state of audit readiness."
Deb G.
Director of Quality, Dimension Therapeutics
"Qualio is far superior to other
systems I've worked with in the past.
The flexibility has been mind-boggling."
Michael H.
QA Director, TriMed
"A game-changer for a paper-heavy QMS. Qualio is a breath of fresh air."
Gareth Q.
Director of Quality, iMIX
"Document control took anywhere from 5 hours to 3 days. Now things get done in hours, even minutes. I can send training out in seconds now. Qualio has made my life 100% better!"
Angela P.
QA Manager, Akadeum Life Sciences
"Qualio helps me sleep better at night.
Having that structure for everyone to work within is essential for us to really succeed this year in getting onto the market and staying compliant."
Heather U.
CEO, EvoEndo
"We passed our MDSAP recertification audit with Qualio!"
John P.
Quality Assurance Manager, KUBTEC Medical Imaging
"Qualio has been great to work with. We implemented an entire QMS with them! Intuitive functionality, friendly staff."
Kim K.
Executive VP of Operations, VirTrial
"I feel 100% ready in our 510(k) and ISO 13485 preparations.
Qualio let us bring quality in-house in a way that will make us successful."
Hilary B.
VP of Regulatory & Quality, SimBioSys
"With other vendors, we felt like just a number.
But this is like a true partnership. Qualio has definitely changed our world."
Eric D.
Director of Quality Assurance, Expression Therapeutics
“We’re building a quality product. So we bought a tool that gives us a quality culture. Instead of pushing
quality to one department, Qualio breaks down that barrier.”
Daniel A.
Chief Technology Officer, InVivo Bionics
"When you're using paper, you're not moving forward. You can't.
Now we're so streamlined, we're finally making progress. We're talking about bringing a new product into design controls right now.
I don't think that'd be possible if we didn't have Qualio."
Ami A.
Director of Ops & Quality Management, NeuFit
"I’ve never been more confident in my 27 years of being audited."
Lowell H.
Director of Quality Assurance, Restech
