SUPPLIER QUALITY
Full visibility into your supply chain compliance
Build and enforce a compliant supply chain. Qualify suppliers, track performance, manage audits, and connect supplier quality to your broader compliance posture.

Intuitive supply chain quality management software
Challenge
Modern supplier quality management demands strict diligence.
But drawn-out qualification, uncontrolled records and patchy visibility all weaken your supplier relationships, impair your control and throw open the doors to third-party risk.
That’s where supplier quality management tools come in.
Solution
Qualio’s supplier quality management software offers a central digital database for categorizing and continuously controlling how suppliers interact with your business and QMS.
Mandate documents, set audit cadences and enforce risk-based policies to give yourself complete oversight of your supplier management system.
Supplier management tools to enforce compliance
- Configure bespoke policies to structure your supplier management activity
- Enforce document requirements and audit frequencies
- Categorize suppliers by risk and govern your supplier activities accordingly
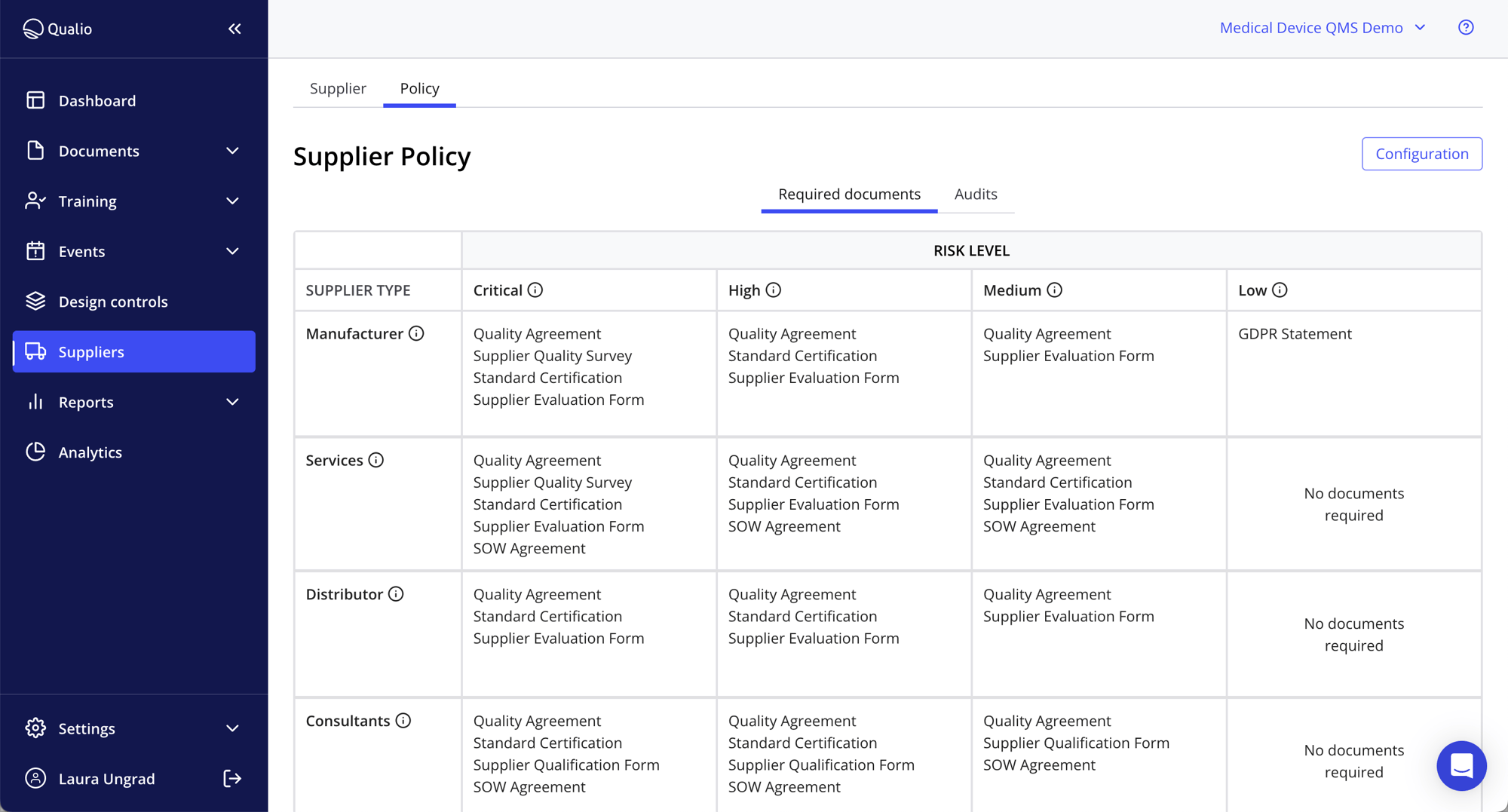
Guide your supplier management system with confidence
- Construct an incorruptible supplier database with key information and data filters
- Add, review, reject and approve suppliers on the fly
- Export an Approved Supplier List (ASL) at the touch of a button
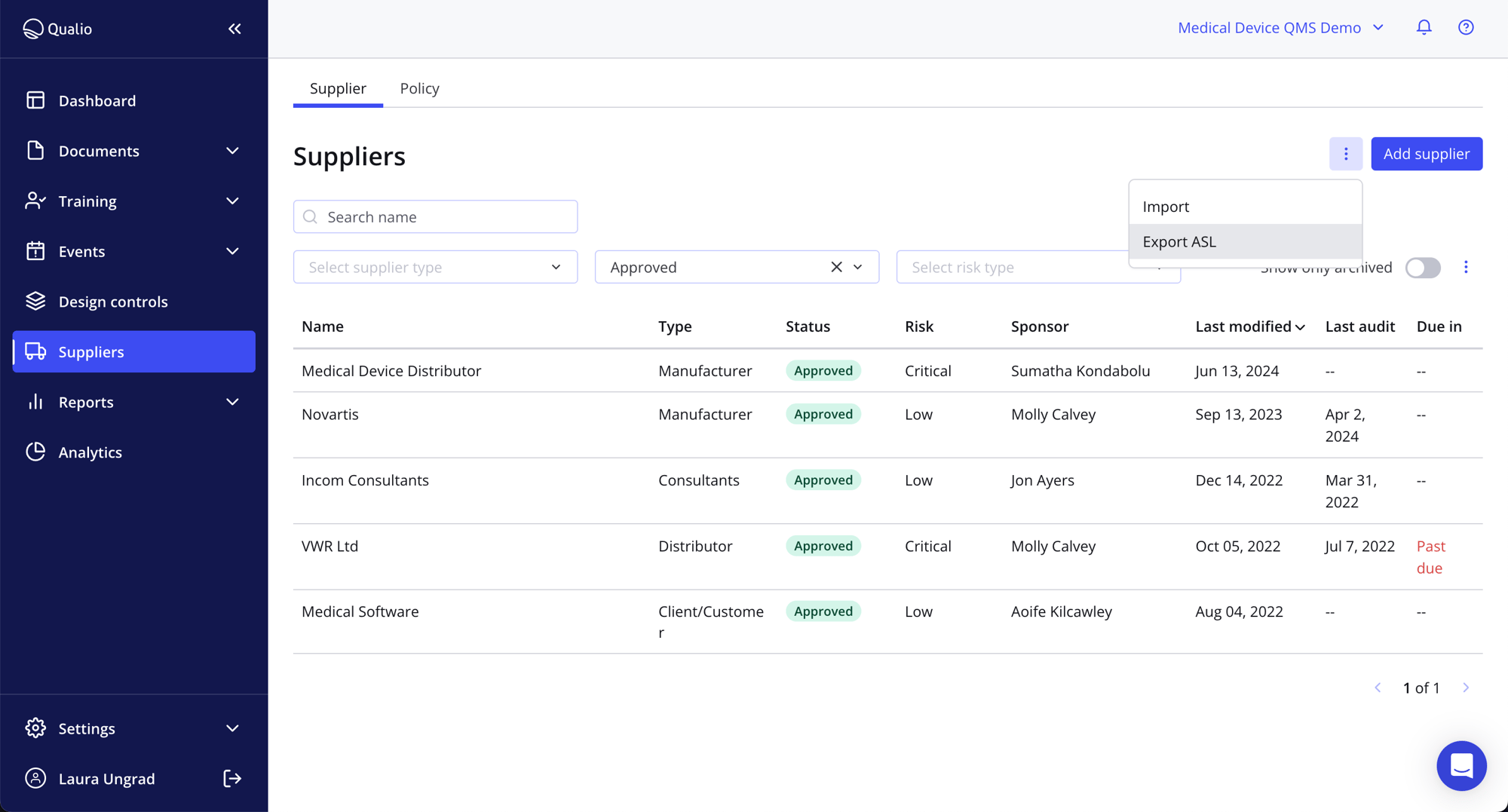
Be constantly compliant
- Meet the requirements of ISO 13485 §7.4, FDA 21 CFR 820.50, ICH Q10 §2.7 and more
- Swap uncontrolled spreadsheets for purpose-built, fully compliant supplier management software
- Onboard suppliers with controlled review and approval workflows
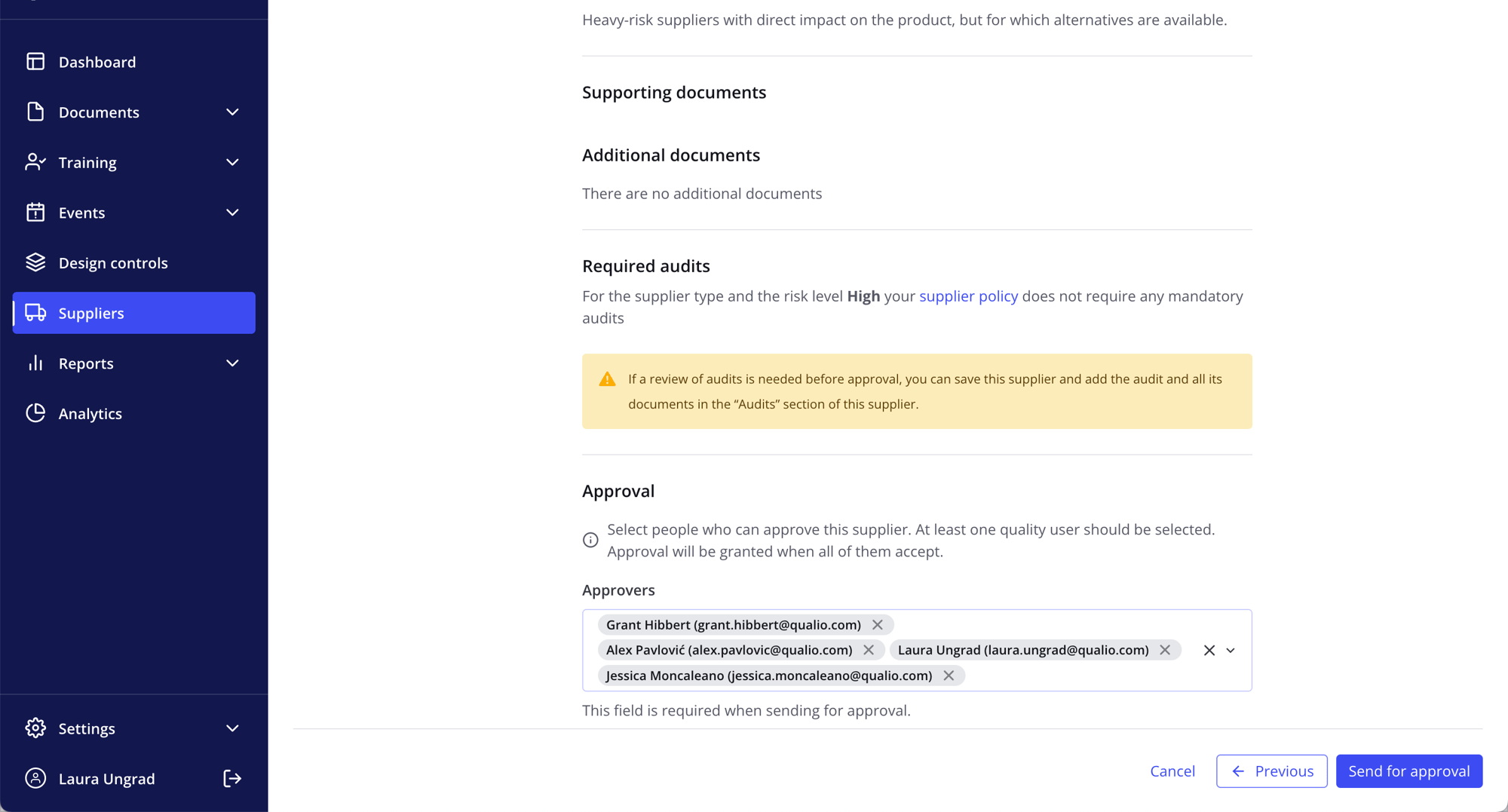
Manage suppliers with speed, ease and accuracy
- Automate third-party control with system-enforced policies
- Slice duplication and pinpoint info quickly with a single source of supplier management system truth
- Never miss a supplier management activity with prompts and reminders for the right person at the right time
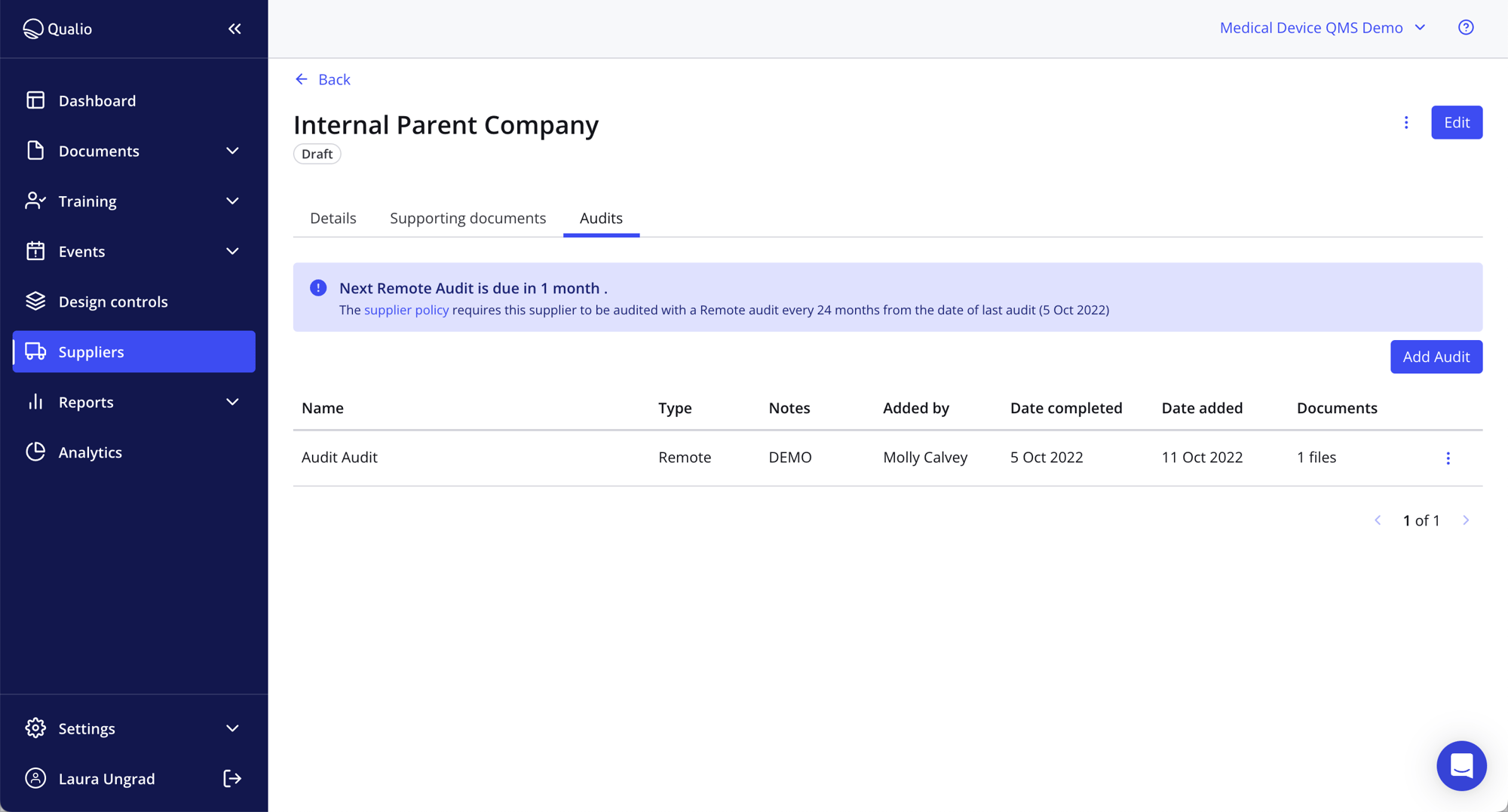
Supplier quality management resources

Essential guide to life science supplier management
Grab total control of your supply chain with our comprehensive best practice guide, from risks and metrics to onboarding and audits.

Supplier management software datasheet
Explore the functionality of our supplier quality management software - and its powerful benefits for your organization.

How to manage your life science suppliers like a pro
Learn common supplier management mistakes and how to avoid them. Explore how to cut risk and maximize supply chain strength.
See the other features that make Qualio
the #1 top-rated supplier compliance software
Document management
Make paper, filing cabinets, folders and spreadsheets a thing of the past.
Design control management
Supercharge your product development from a single source of truth.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Change control management
Manage change activity easily and effectively with complete traceability.
Audit management
Make compliance and audit readiness the natural core of your business DNA.