CE AND UKCA MARKING
CE and UKCA marking for medical devices: open the European and British markets 🇪🇺 🇬🇧
Get your CE and UKCA marks, and keep them.

CE and UKCA marking with ease
Challenge
Medical device and in vitro medical device companies need to prove their products are safe and compliant to secure their CE and UKCA conformity marks.
But complex, shifting legislation and the impact of Brexit have made cracking the EU and UK markets a tricky task.
Manual quality, siloed processes and murky visibility make lasting compliance an onerous, time-consuming and energy-sapping burden.
Solution
Qualio's fully integrated eQMS software arms medical device companies with the tools they need for confident, long-term adherence to EU MDR/IVDR and UK MDR requirements.
Automatic, AI-powered gap analysis and a cloud-based single source of truth make it easy to connect your workforce to a collaborative, compliance-focused culture.
CE and UKCA marks become a natural byproduct, not a stressful target.
Used by 700+ companies for compliance with...

CE and UKCA marking software: how it works
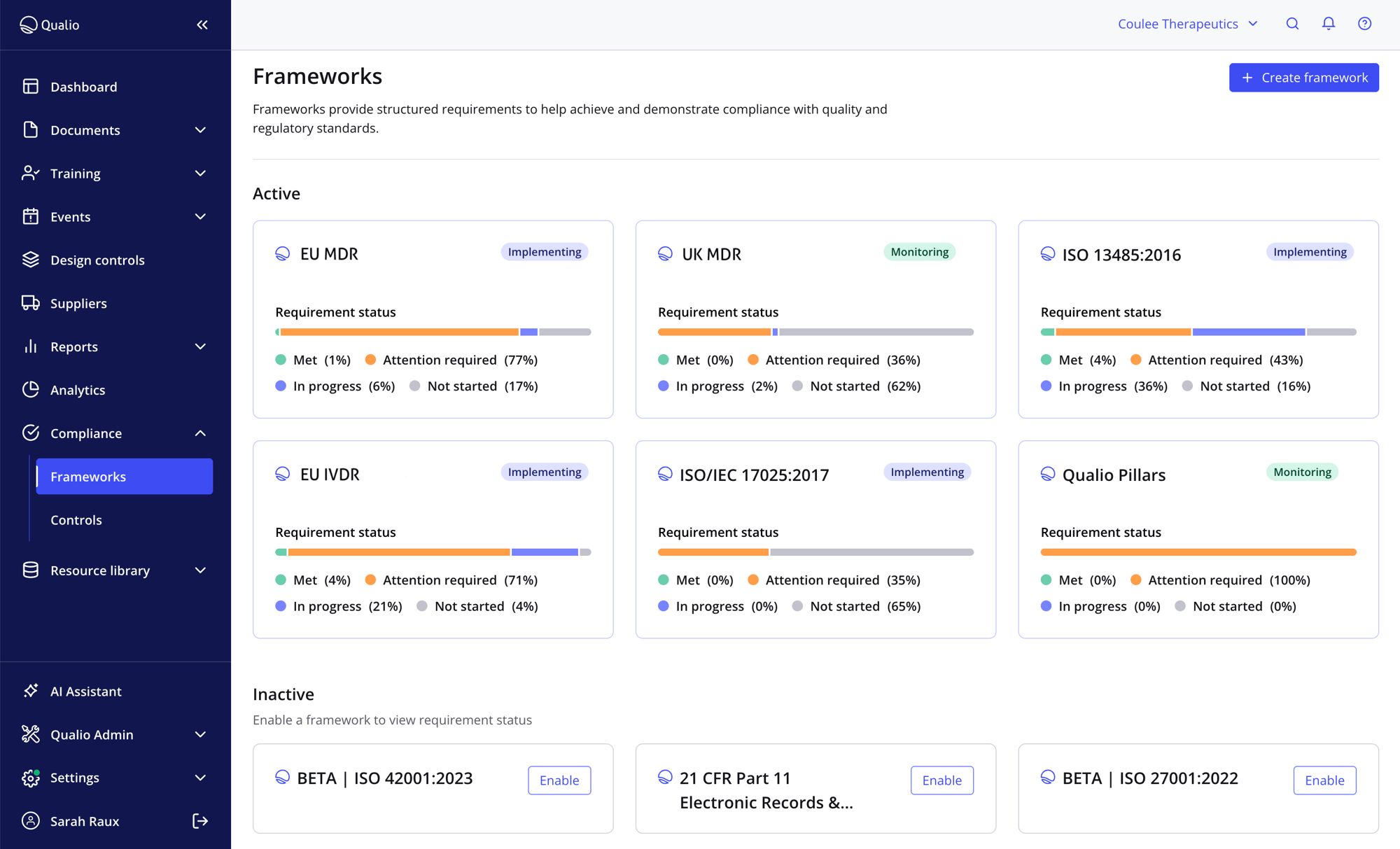
Total EU & MHRA audit readiness
- Automated gap analysis scans your QMS in 30-40 minutes, highlighting every EU and MHRA compliance gap in your organization
- Allocate tasks and automatically gather device evidence to get audit-ready in weeks, not months
- Enjoy total CE and UKCA marking readiness with constant monitoring and compliance health dashboards
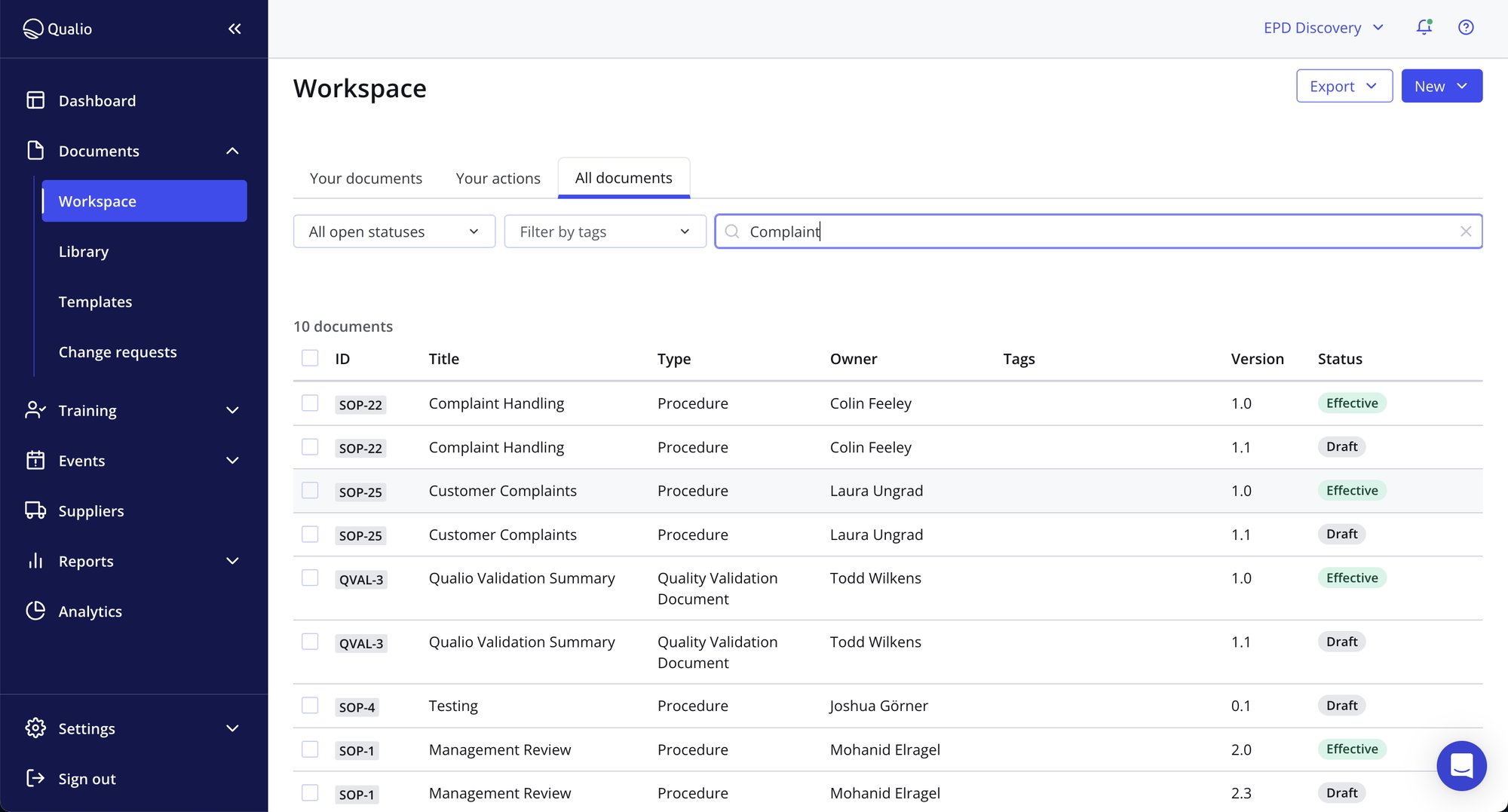
Integrated, best-in-class medical device eQMS
- Access a ready-made structure for compliance, from design controls and management responsibility to clinical evaluation and UDI
- Meet every single marking requirement by aligning your teams around version-controlled documents, training records, workflow-driven digital processes and more
- Make your Notified and Approved Body auditor happy with instant access to centralized, audit-trailed and inspection-ready quality data
CE and UKCA marking medical device resources

EU MDR software datasheet
Dive deeper into how our software empowers your business for lasting EU market presence.
EU IVDR software datasheet
Bringing an IVDR to the EU market?
See how Qualio gives you everything you need.

CE marking pathway guide
Access a step-by-step pathway guide for securing a UKCA mark.

UKCA marking pathway guide
Access a step-by-step pathway guide for securing a UKCA mark.