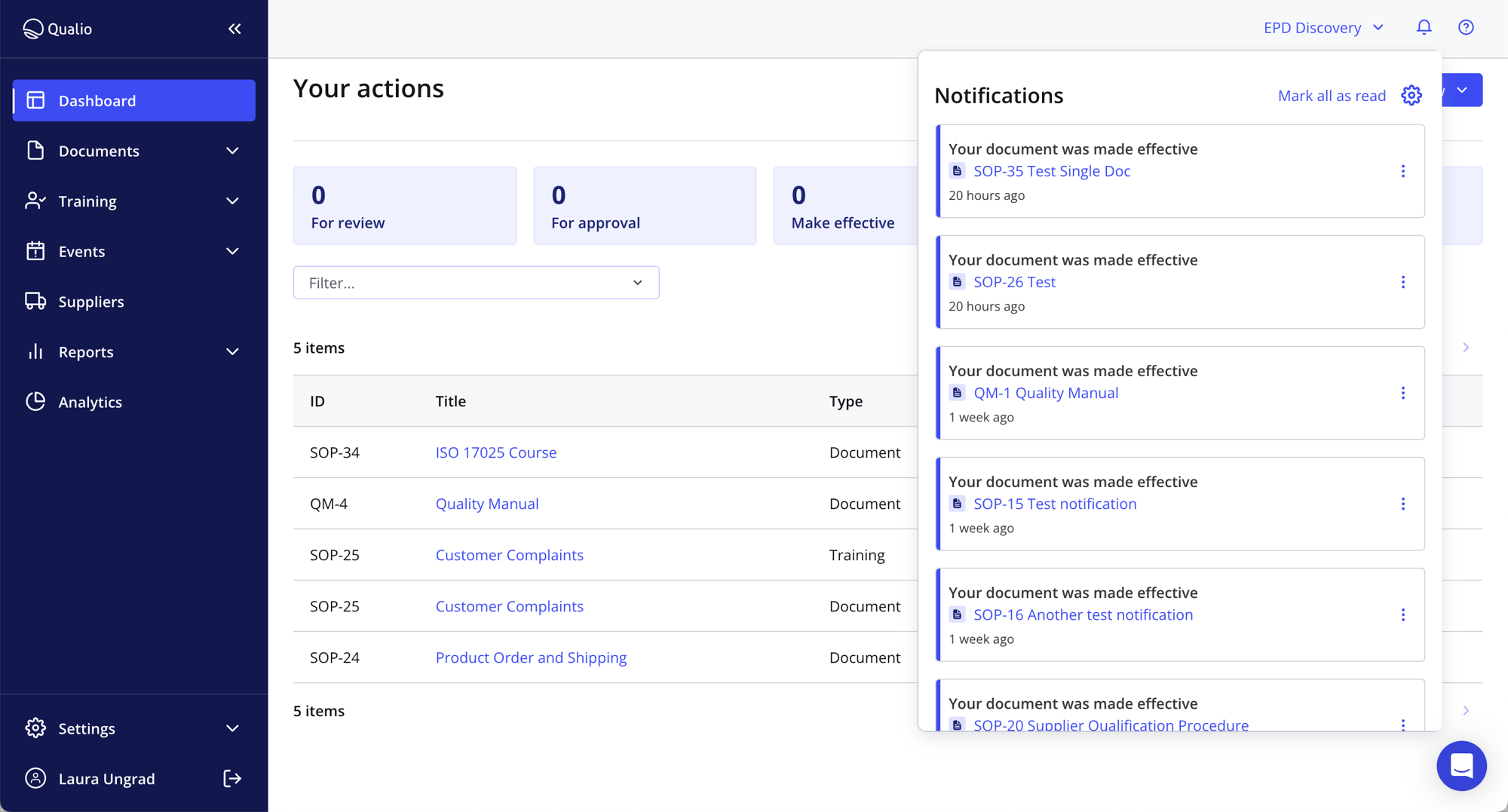
FROM PAPER TO PLATFORM
From paper to platform
Cut the clutter, chaos, and compliance complications. Move to a modern compliance platform that your team will actually use.

A fully paperless QMS approach
Challenge
42% of life science businesses still rely entirely on paper for their quality efforts.
Paper-based quality systems slow work, block visibility and breed risk.
From document version control to proving competence and tracing actions, your paper QMS quickly breaks down in the face of regulatory scrutiny and holds your business back from its true potential.
Solution
Qualio provides an integrated, cloud-powered and fully digital paperless QMS software system that makes paper a thing of the past.
Electronic documents, records, workflows and e-signatures embed complete control and visibility into the heart of your quality system.
Actions accelerate, compliance strengthens, insights sharpen, and quality becomes an agile tool for continuous improvement.
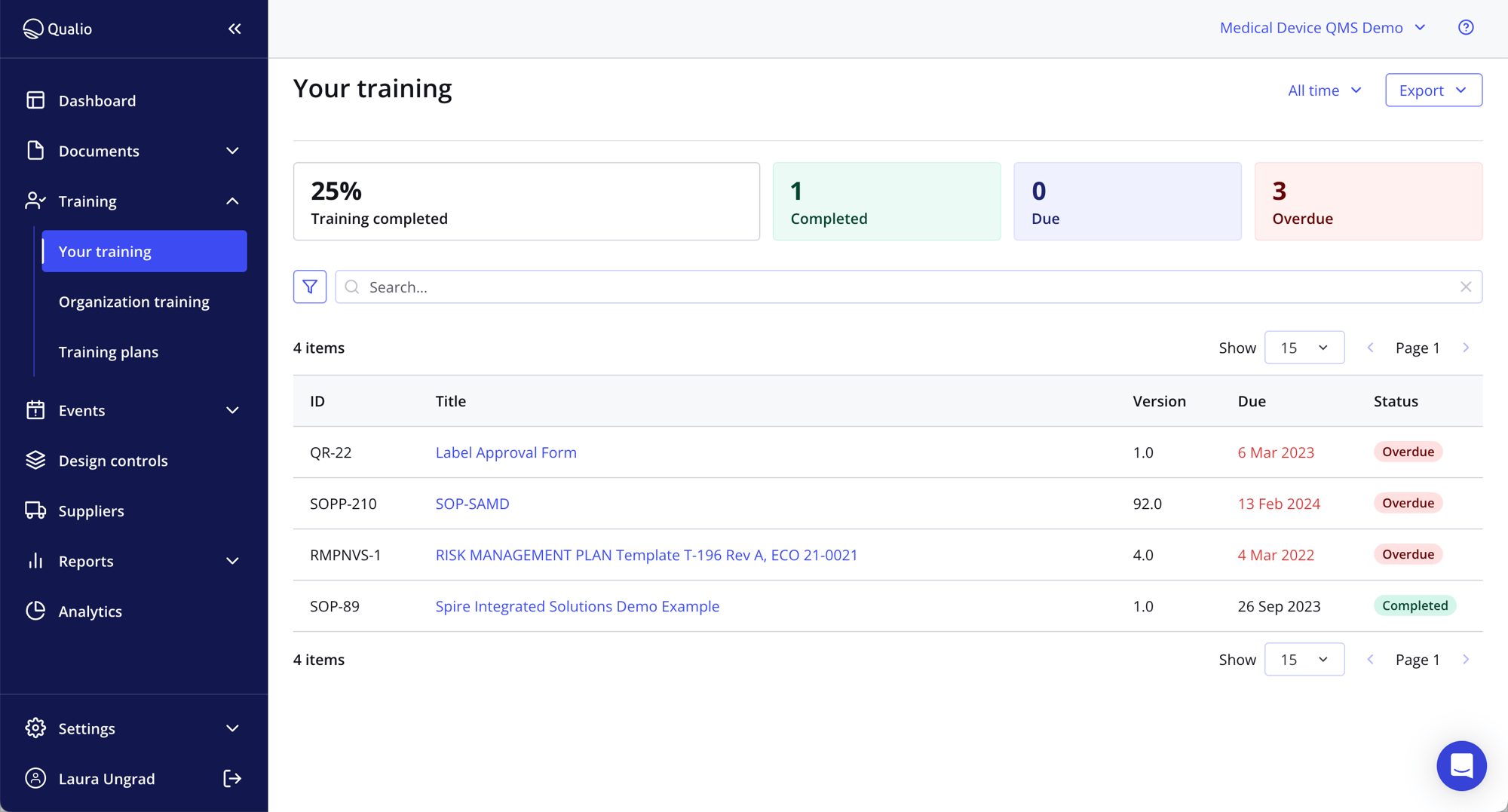
A single source of truth at your fingertips
- Get back the 30 minutes each of your colleagues spends every single day looking for paper documents
- Access info in seconds from a single paperless document management system
- Handle documents, training records, event workflows, suppliers and design controls with ease and without duplication
Lift the lid on growth
- Replace paper admin time with value-add time
- Build a culture of quality underpinned by real-time digital data
- Redirect the time, energy and clutter of a paper system into agile, connected work
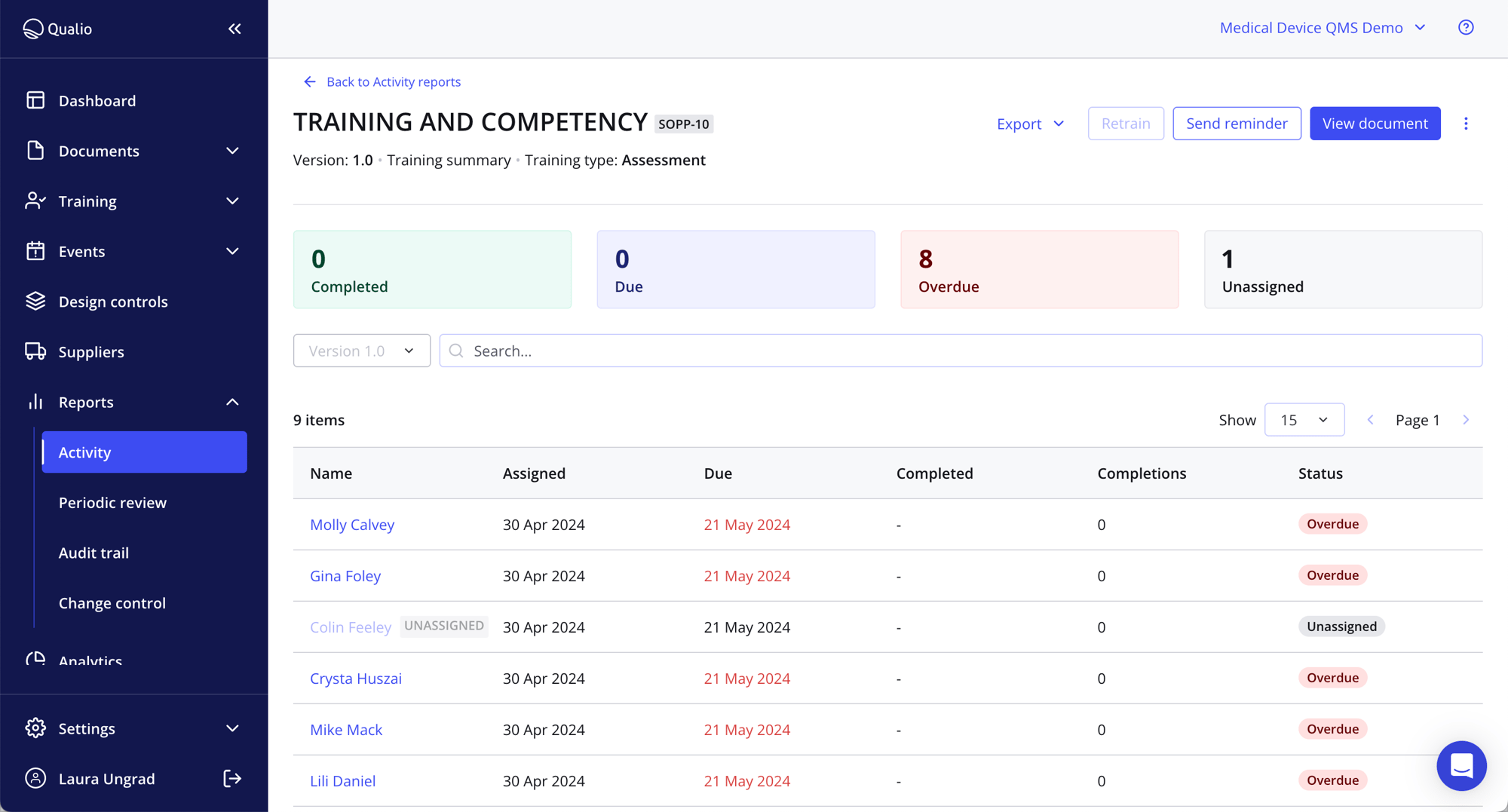
Cut costs
- Jettison hard document production, usage and storage costs (AIIM estimate: $25,000 a year minimum!)
- Get to market and generate revenue quicker with accelerated, digitized processes
- Make your QMS leaner, sharper and less labor-intensive
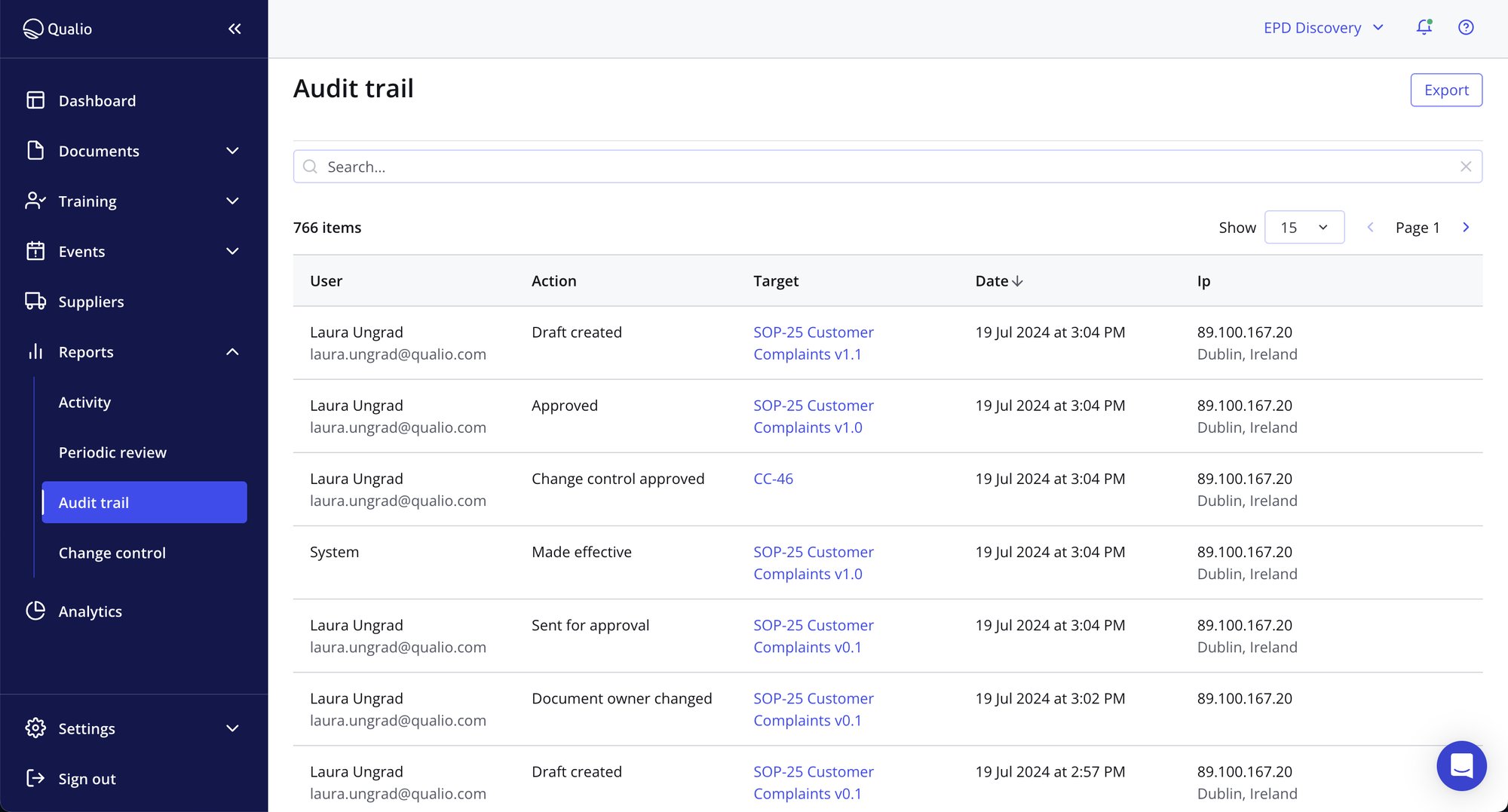
Get confidence and control
- Make GDocP, version control and ALCOA+ effortless and automatic
- Be prepared for any audit — without the filing cabinet stress
- Replace desk-to-desk wet signatures with cloud-based, fully compliant e-signatures
Keep up with industry expectations
- Align with the digital best practice in the FDA's Case for Quality and QMM programs
- Answer regulatory demands for optimal, digital quality management
- Prove your business is a modern, quality-centric operation
Paperless QMS resources
Qualio vs. paper: functional comparison
Compare our paperless document management software with a paper-based QMS, from costs and processes to scalability and usage.

The case for electronic document management
Learn the 6 key reasons your life science business should embrace a paperless document management system - and the powerful benefits you can expect.
How paperless document management software changed everything for a Qualio customer