Complete guide to computerized system compliance
Understand everything you need to know about modern computerized system adoption, from validation to vendor best practice.
Instant access. No email required.
Download our computerized system compliance PDF guide to:
- Understand the evolution from computerized system validation (CSV) to assurance (CSA), and what it means for your business
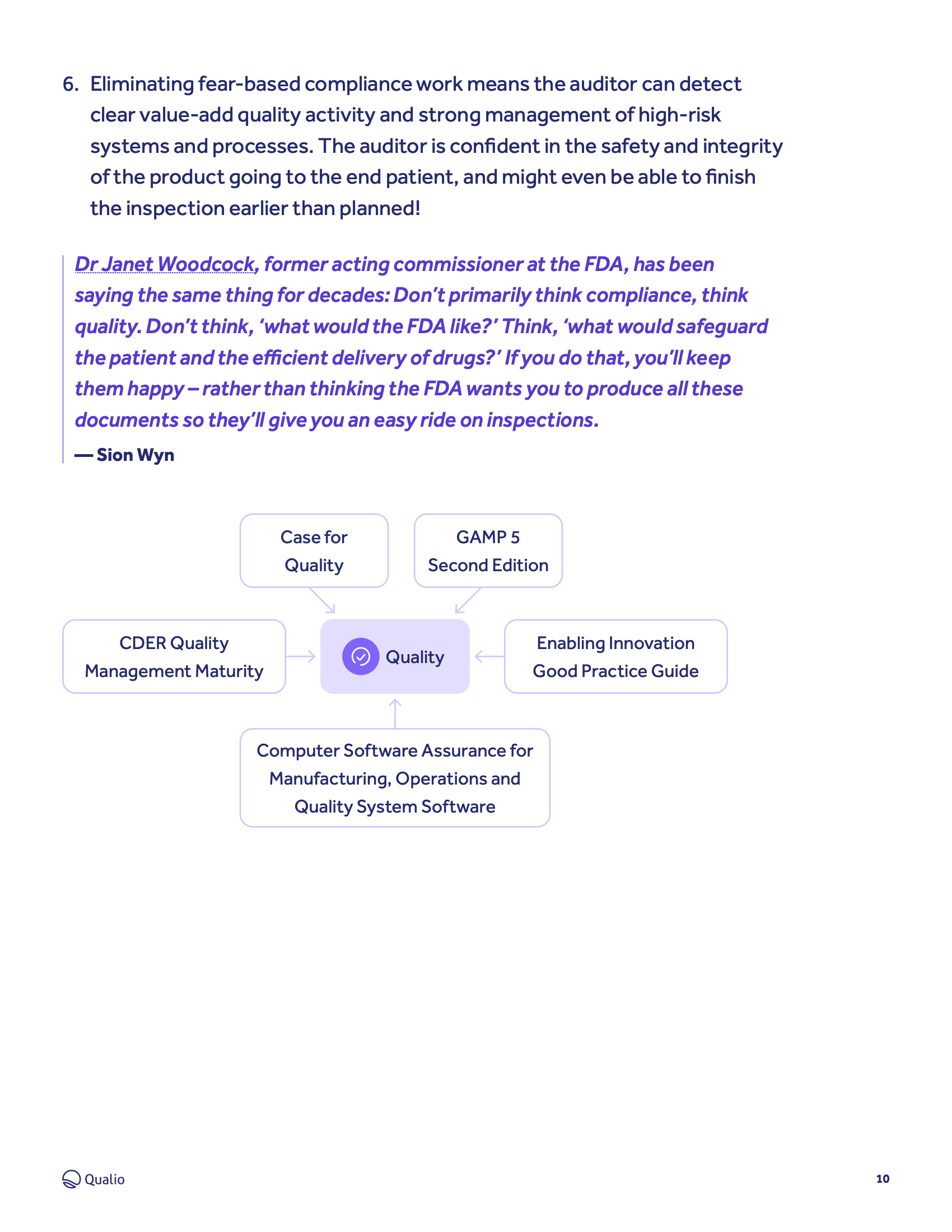
- Access expert breakdown analysis of the GAMP 5 2nd Edition, the Enabling Innovation GPG, and more
- Learn how to confidently adopt digital tools that strengthen your life science business and satisfy your auditors
What you'll get:
Expert guidance
We sat down with Sion Wyn, editor of GAMP 5 and advisor to the FDA, for his insight, tips and commentary on the world of modern computerized system compliance
Validation demystified
What does modern adoption of computer systems demand? What are regulators really looking for? Do you still need traditional documents like IQs, OQs and PQs? Answer these questions and more
Roadmap to success
Learn the common errors that regulated businesses make with their adoption of computerized systems, and how to avoid them