POSTMARKET SURVEILLANCE
Continuous postmarket monitoring and market oversight
Full visibility across your product's market journey. Protect drug and device safety with connected postmarket surveillance.

Complaint management software tools for long-term market success
Challenge
Getting your drug or device to market is only the beginning. Hundreds of life sciences products are recalled annually, resulting in hundreds of millions of dollars of revenue loss.
Complaints, adverse effects, feedback and continuous improvement opportunities all need to be monitored and acted on.
Disconnected manual tools block your visibility, hide trends, and make a full picture of market performance impossible.
Solution
Qualio's postmarket surveillance and pharmacovigilance software gives your business a connected web of information for snappy, data-driven postmarket activities.
Build market triggers to kick off automated quality management workflows.
Track every quality event and issue by product.
And use detailed analytics to spot and address trends before they become harmful.
Instant integration with your third-party tools
- Connect Qualio to your CRM and ticketing systems, such as Salesforce, Zendesk, Intercom or HubSpot
- Automatically trigger complaint management software workflows in Qualio as postmarket complaints are logged elsewhere
- Drive closed-loop actions to completion to keep your product compliant and your customers happy

Connected quality data to knit your postmarket activity together
- Build, collaborate on and distribute key PMS and pharmacovigilance documents like SOPs, CERs, RMPs and PSURs in a single source of truth
- Connect product data using system links and centralized data points like tickets and customers
- Pull tickets from design tools like Jira and GitHub into Qualio to inform design updates and continuous improvement
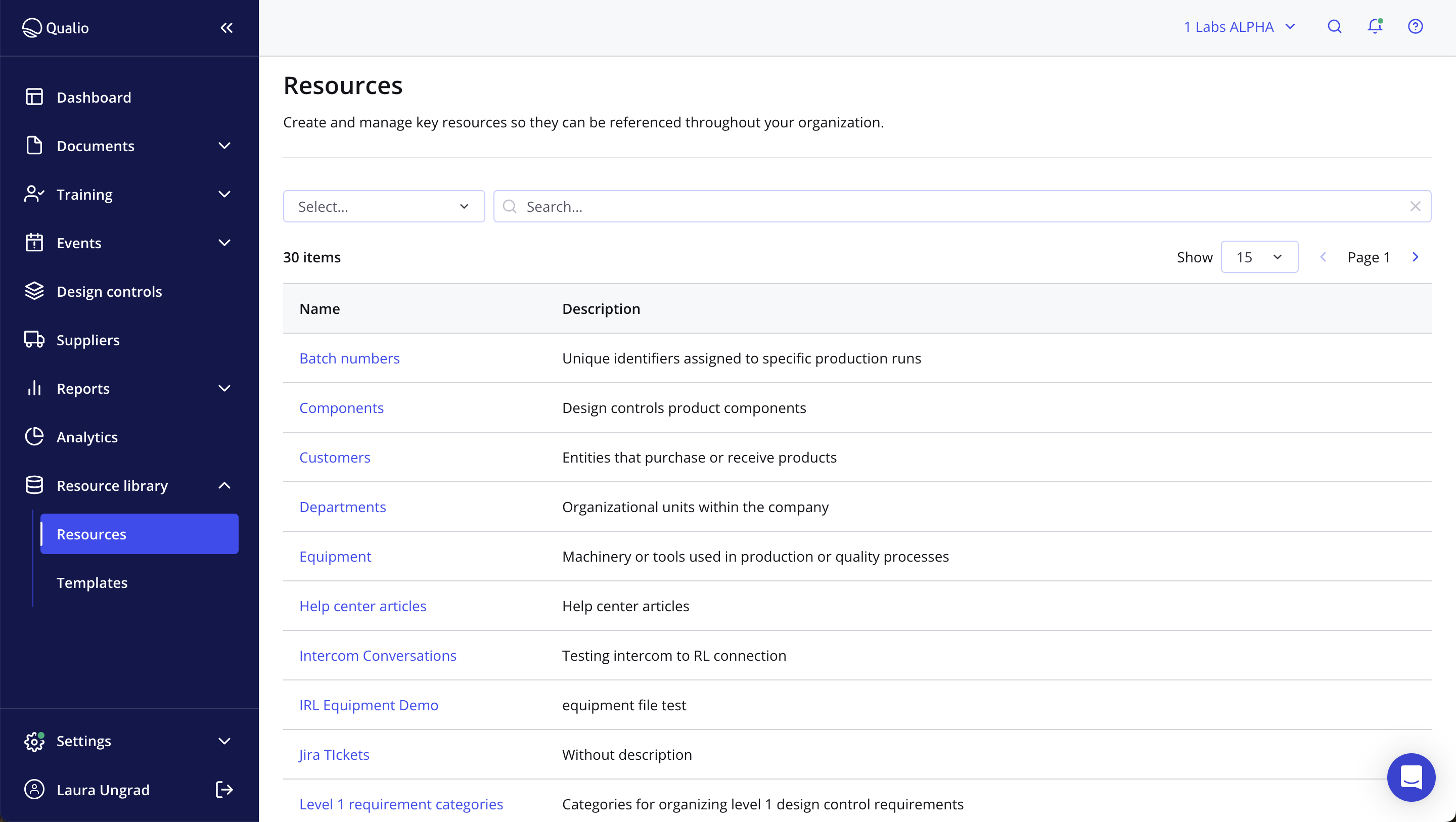
Take smarter action for optimal product quality
- Respond to market events in real time with automatic prompts and reminders
- Shape bespoke workflows to collaboratively manage adverse effect investigations, CAPAs, health authority notifications and more
- Pull key data like batch numbers, suppliers, materials and departments into forms and workflow actions for total traceability
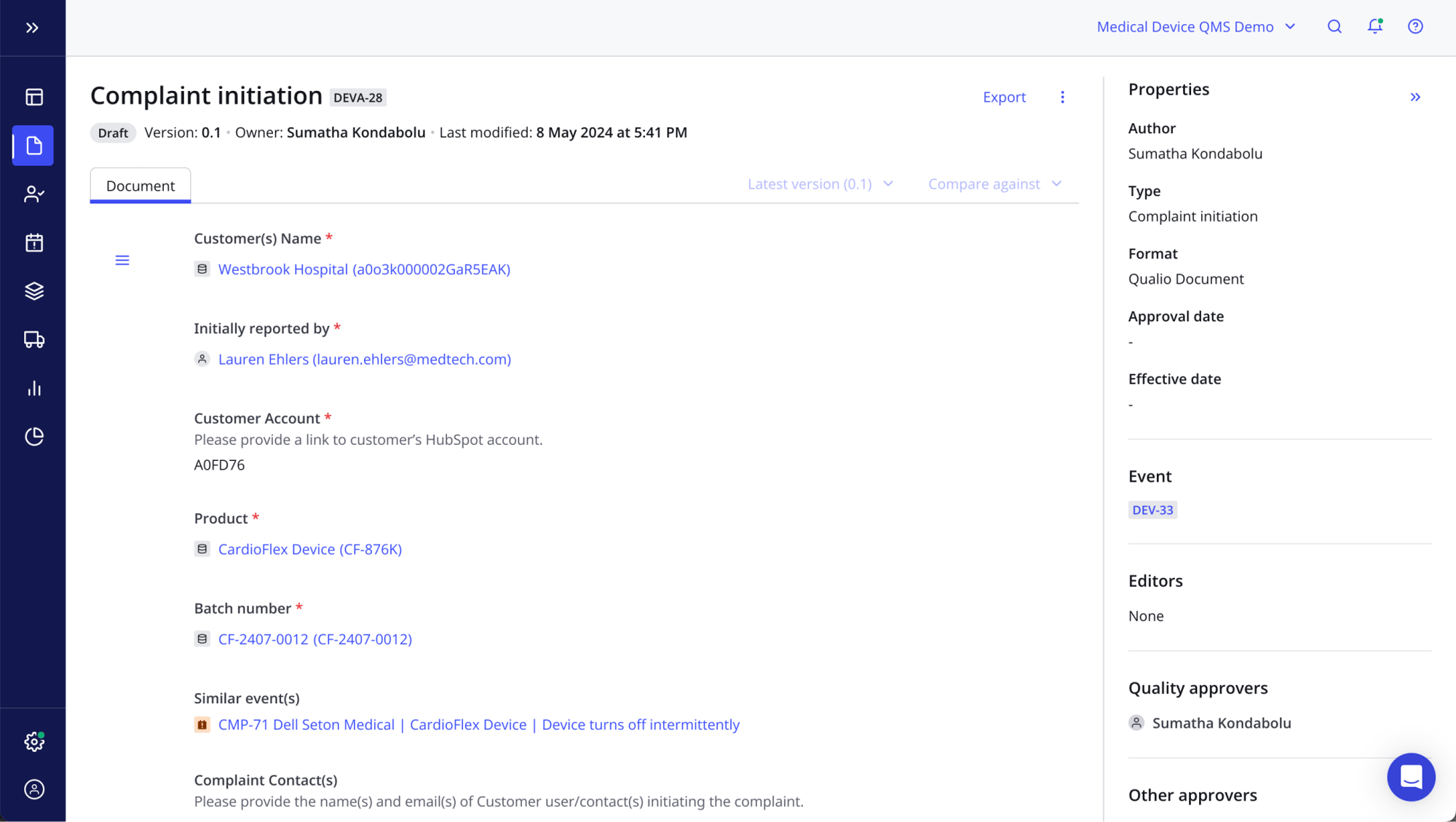
Get the product insights you need
- View detailed quality histories by product, from past CAPA actions to design updates, procedural changes and non-conformance records
- Spot product trends and weaknesses early
- Demonstrate total control and traceability to your auditors and inspectors
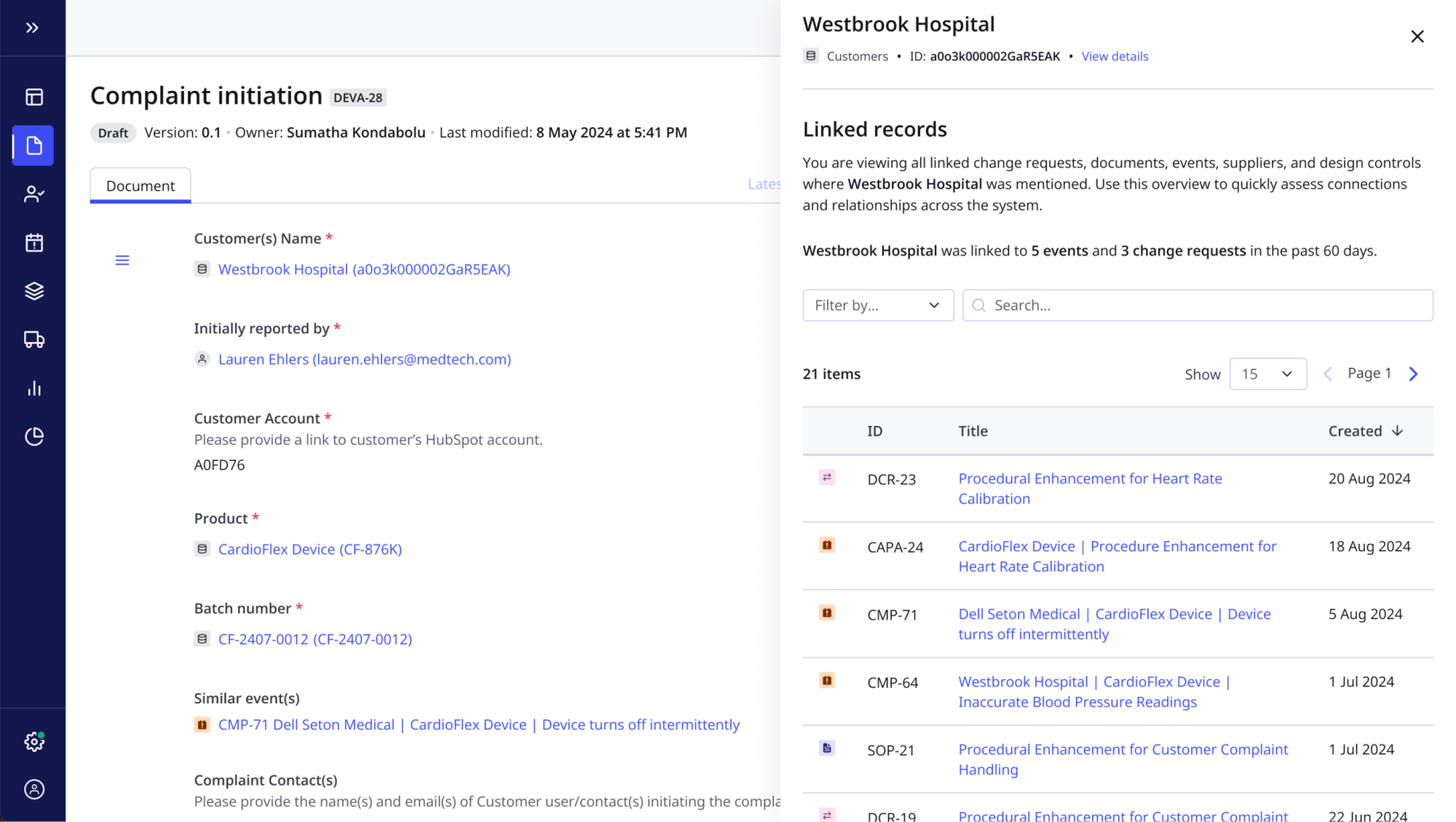
Postmarket surveillance & pharmacovigilance resources
Understanding post-market surveillance for medical devices
Read our blog post guide to understand the key operational ingredients of modern post-market surveillance activity.

Medtech post-market essentials webinar recording
Catch our webinar recording for an expert-led walkthrough of the key postmarket requirements your medical device company needs to meet
Not just complaint management software...
Design control management
Supercharge your product development from a single source of truth.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Change control management
Manage change activity easily and effectively with complete traceability.
Audit management
Make compliance and audit readiness the natural core of your business DNA.
Supplier management
Control and categorize suppliers and third parties with complete visibility.