From treading water to real quality improvement
Who are NeuFit?
Headquarters
—Austin, TX
Company size
—10-50
Industry
—Medical Device
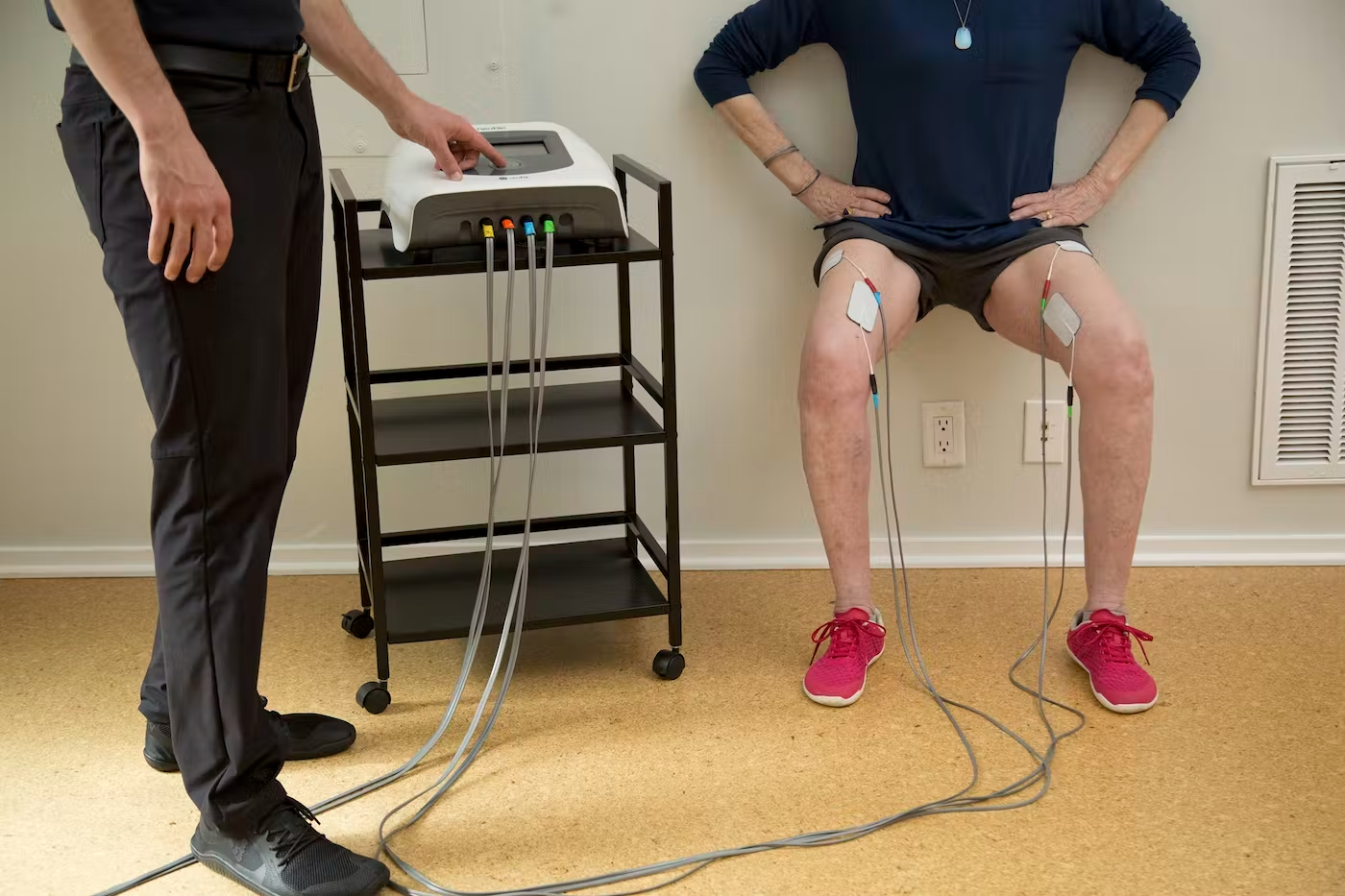
NeuFit is the Austin-based manufacturer of Neubie, a neuromuscular electrical stimulation medical device.
Neubie helps patients recover from neuromuscular damage, and can treat conditions from plantar fasciitis to strokes and spinal cord injuries.
NeuFit must work to the requirements of ISO 9001, ISO 13485 and ISO 14971.
The challenge
NeuFit relied on a manual paper- and Dropbox-based system to manage their quality activities.
Director of Operations & Quality Management Ami Anderson spent 'every day, all day, and a lot of overtime thrown in' just completing quality admin tasks.
And this excessive quality timesink meant the other half of her role - operations - didn't get the same attention, causing issues in areas like supplier management.
The issues with NeuFit's manual system came to light when an FDA auditor arrived:
Our inspector let me know she was really upset with how we were managing everything.
It took us a long time to get the documents she wanted. We had to make copies because she couldn't take the originals.
She'd expected to be able to load a thumb drive and didn't have a bag big enough for everything. And I wasn't allowed to give her a bigger one!
Despite having limited experience of digital quality management, Ami knew an eQMS would strengthen NeuFit's audit readiness and unlock stronger, sharper processes that would save her time.
The solution
Ami homed in on three potential eQMS vendors for closer inspection.
One option still required lots of manual change control, including downloading system documents for revision then re-uploading.
The second option put Ami off with overly pushy sales tactics, which included putting down other eQMS vendors and over-promising functionality.
During discussions with Qualio, Ami felt comfortable to discuss and explore her operational requirements without pressure. On top of that, the system itself met all of NeuFit's needs.
After NeuFit's CEO Garrett Salpeter was impressed by Qualio too, sign-off was given to become Qualio customers.
The results
Replacing NeuFit's manual QMS with Qualio bore fruit immediately.
The need to invest in additional headcount and consultancy was removed, saving NeuFit $130,000 in planned spend.
The visibility and control of an eQMS allowed Ami to spot and fix quality issues invisible in a paper system, resulting in a boost in customer satisfaction and allowing a failure part to be detected and fixed early.
Quality events are closed out more quickly, and the organization anticipates an accelerated route to market for their next medical device.
And best of all, the 6-day events of regulatory audits can now be completed in half a day.
After the disappointment of their previous FDA inspection, Ami is confident, happy and excited to show their new quality system off next time:
We're so streamlined now.
It's amazing where we were, compared to where we are now.
I hope we get the same auditor in future, so I can say, 'look how wonderful this is now!'
Hear from Ami
SEE QUALIO FOR YOURSELF
We won't pressure you either!
See what Ami liked about talking to us.