MEDICAL DEVICE COMPLIANCE
Medical device compliance, automated
Design controls, risk management, and multi-framework compliance in one platform. AI agents handle gap analysis and regulatory monitoring so your team ships devices.

Medical device quality management system software for modern challenges
Challenge
Many medical device companies work across multiple systems that create a siloed approach to quality management.
Some have a home-brewed CAPA approach, or a separate tool for design controls. Others are stuck in paper-based systems that clutter and complicate quality.
These approaches don't scale. And they slow down the release of the innovative, life-saving products your patients need.
Solution
Qualio replaces your legacy systems with centralized, cloud-based medical device QMS software that's accessible from anywhere.
Flexible workflows and templates give you room to mature and scale your quality processes with your business.
And design controls, documents, training and more are all handled in a consistent way - all underpinned by powerful reporting and audit trailing for complete visibility.
Medical device quality management system software for prototype to production
- Guarantee quality with easy-to use QMS software for medical devices that grows with your company
- Digitize your document stack and share information quickly and easily
- Execute workflows for document approval, design controls, training and more
- Manage suppliers, audits and change control across your entire business in real time
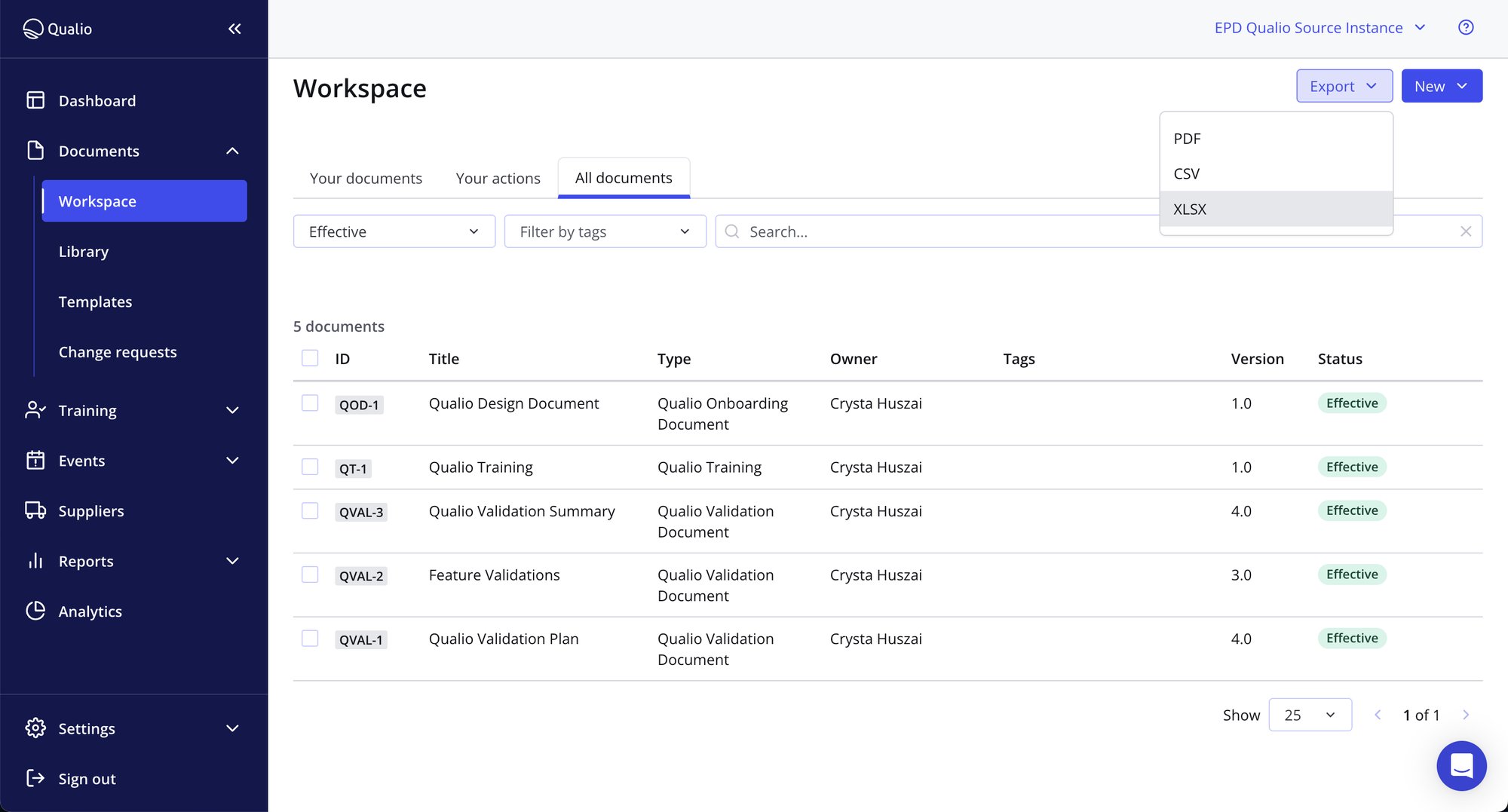
Have confidence in your medical device QMS
- Unlock complete visibility of your medical device QMS landscape, from design controls to outstanding training
- Pull design elements from Jira, TestRail and more straight into Qualio
- Find the information you need in seconds from a single source of truth
- Generate release documentation, from ISO 14971 risk assessments to user requirements and Change History Reports, with a single click
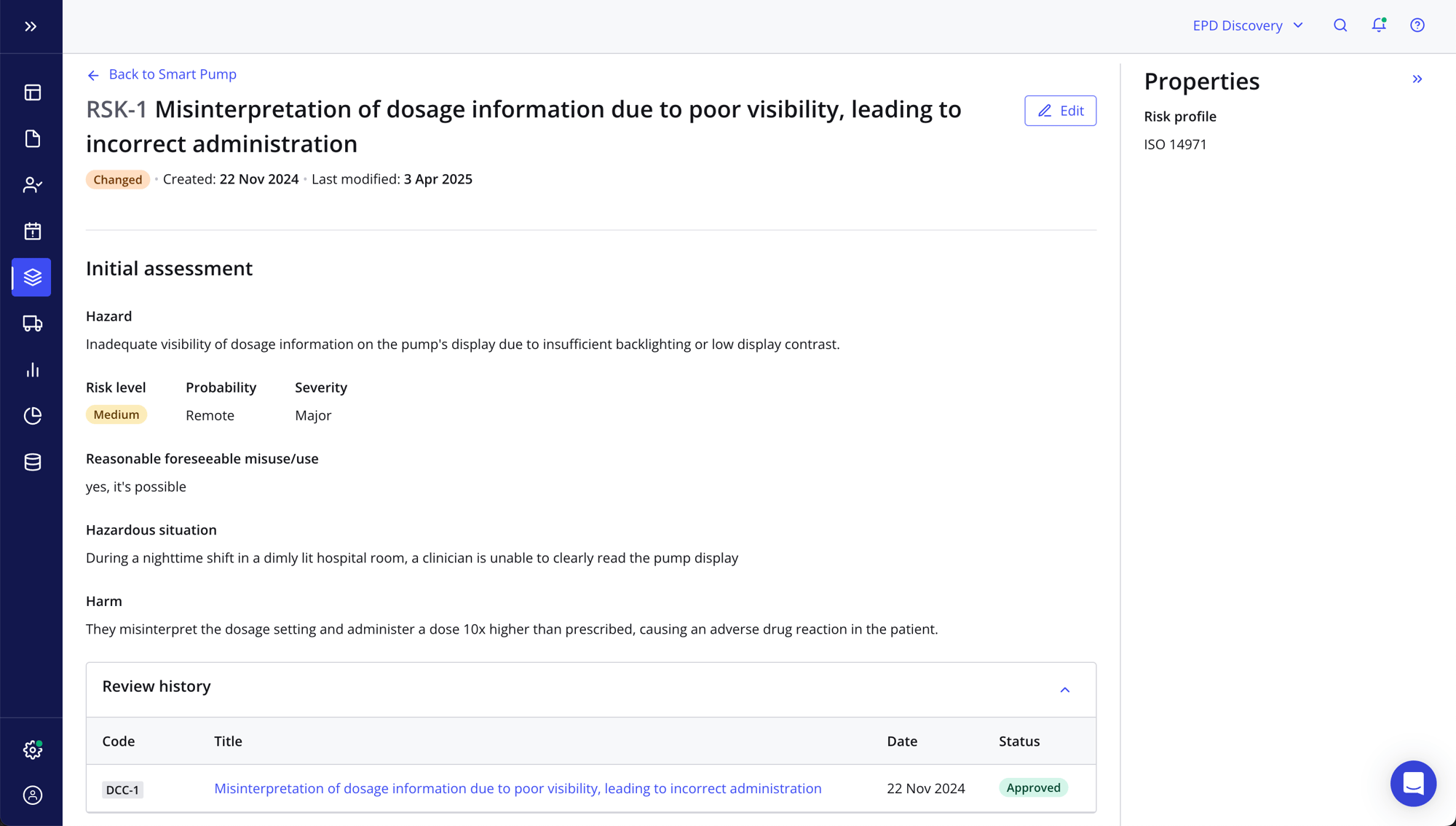
Successfully navigate complex regulations
- Easily comply with ISO 13485, ISO 14971, the MDSAP, FDA 21 CFR Part 11/820 and more
- Seamlessly weave risk management into your design and development activities for a right-first-time approach
- Build a clear audit trail and end-to-end traceability for all product information
- Get expert advice from our best-in-class quality advisors
Medical device QMS resources
Medical device quality management software datasheet
Learn how Qualio arms your business with a holistic, compliant medical device eQMS - from design controls to document management

ISO 13485 toolkit
Download our toolkit to access a range of ISO 13485 resources.
Get a clause-by-clause compliance checklist, an audit readiness guide, a design controls e-book, a certification guide and more
Trusted by medical device companies worldwide
Hear from our customers
"I would recommend Qualio to small companies implementing a quality system for the first time. Qualio helped us reduce our workload and our paper trail."
Paul M.
CEO, Arann healthcare
“We evaluated all players on the market and came up with Qualio as the best balance of price, capability, and quality of support.”
Jeffrey B.
CFO, FifthEye
“Qualio enabled us to seamlessly work through our ISO 13485:2016 requirements and design errors out of our quality management system. One way Qualio achieves this is to ensure that changes flow through our system and are applied globally.”
David H.
CEO, Surfatek
"Qualio+ fits right in to ensure a smooth transition to an eQMS. The team offered excellent advice every step of the way."
Chris S.
Quality Manager, CureMetrix
”A digital trail is so important in medical devices, particularly around field servicing and updating. It's just lovely to know that you have these automatic links and alerts, and that two years from now when you're looking back, you will find it.“
Andrew L.
CEO, ArcScan
"We passed our MDSAP recertification audit with Qualio!"
John P.
Quality Assurance Manager, KUBTEC Medical Imaging
"Qualio is a superb eQMS solution for medical devices."
Andrew R.
Director of Tech & Quality, Medbaye
"The validation documentation provided by Qualio was easy to understand and execute. I really like the in-app support and chat function."
Soni M.
QA/RA Director, NovoSource
More content for medical
device professionals
'Firing on all cylinders' with a medical device eQMS
Zalgen Labs replaced their manual, paper-based QMS with Qualio - and the results were transformational. Hear the story straight from Director of Diagnostics Development Matt Boisen.
Building a real quality culture with Qualio
QA Manager Angela was snowed under with quality admin passed to her from her colleagues. Qualio cut 99% of her admin time - and 'lit the fire' of a collaborative quality culture with it.
Quality excellence for a medtech consultancy - and their clients
Effectum Medical wanted a best-in-class quality system to enhance their own work, and to share with their regulated start-up client base to get them to market faster. With Qualio, they got both.


