DESIGN CONTROLS
Full traceability from requirements to release
Link requirements to verification to validation in one connected system. Risk matrix, design outputs, and DHF generation with a complete audit trail.

Streamline your device development
with design controls management software
Challenge
Quality teams work in one system. Engineering teams in another.
Siloed and disconnected teams and platforms make traceable, compliant product development impossible to manage.
Work grinds to a halt, quality and development work against each other, and your path to market becomes a complex nightmare.
Solution
Qualio's design control software offers a single source of truth for all your product development activities.
Pull product data into Qualio with integrations. Control inputs, requirements and risks. Bring your quality and engineering teams into sync.
All underpinned by a complete, compliant document stack to show your auditors.
Unite systems and data to enable effective collaboration and stop delays
- Demolish quality and engineering siloes by connecting systems like Jira, Azure DevOps and TestRail to a centralized, compliant medical device design software platform
- Build a real-time, always-up-to-date document stack that flows seamlessly from your engineering team to quality
- Centralize all design control data by product, from custom categories and components to requirements, inputs, outputs, risks and drag-and-drop attachments
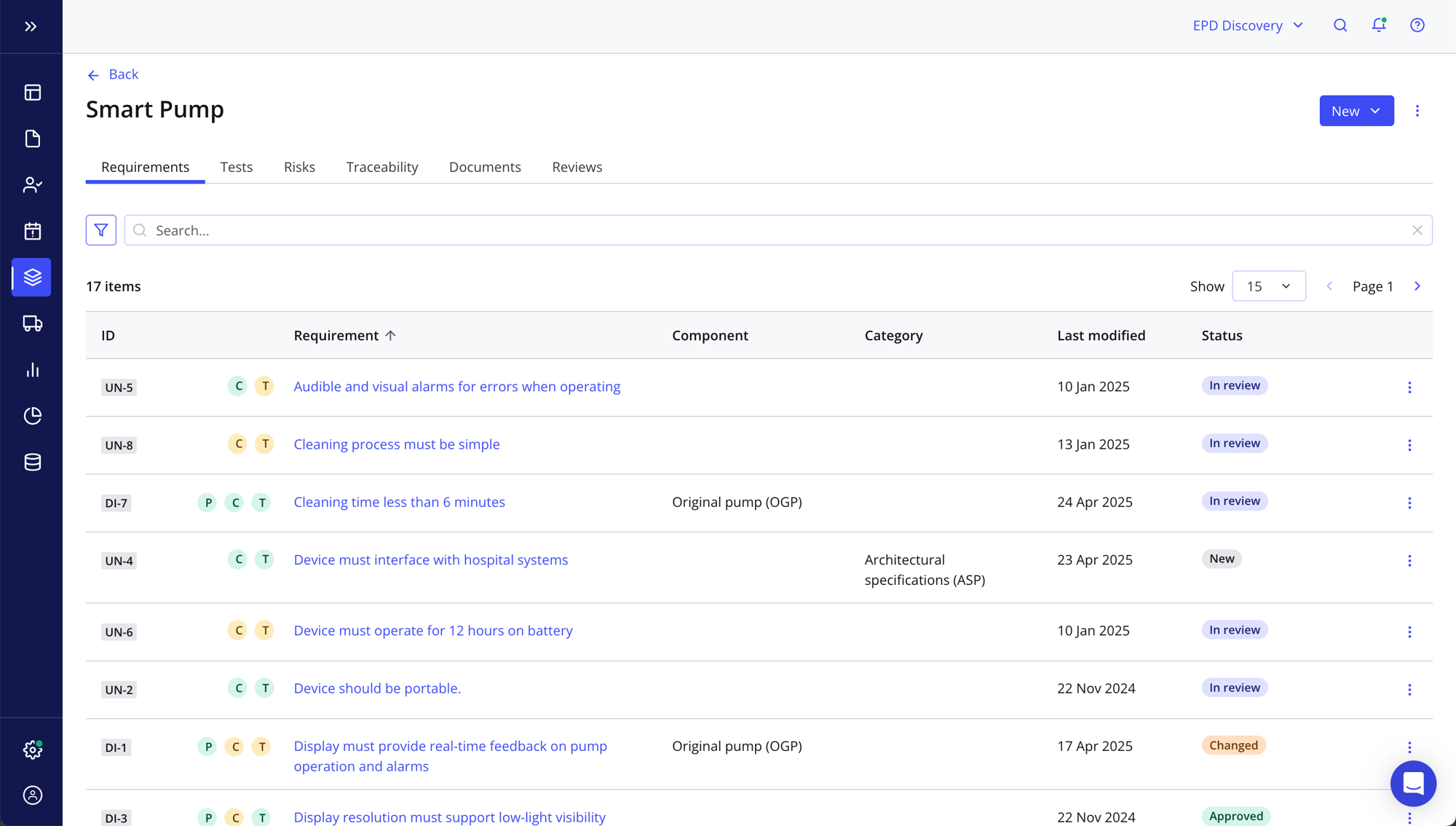
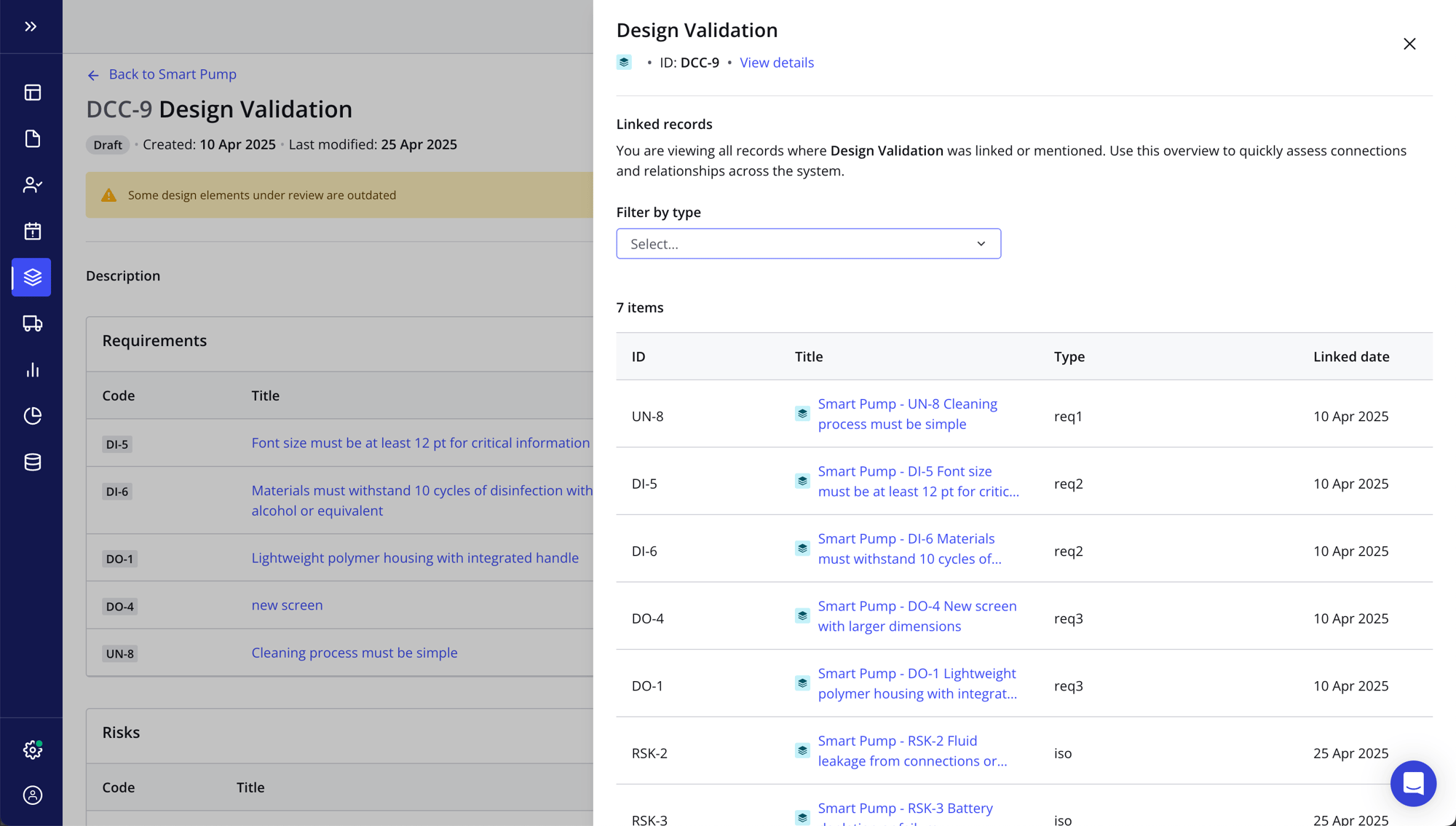
Maintain end-to-end traceability across systems in real time
- Stay totally compliant at all times with automated prompt icons that draw your attention to where it's needed, from missing connections and unresolved risks to outdated test results
- Drive changes and product updates quickly and easily
- Use the Traceability tab for a bird's-eye view of your design elements while your engineering teams work in the source systems they know and love
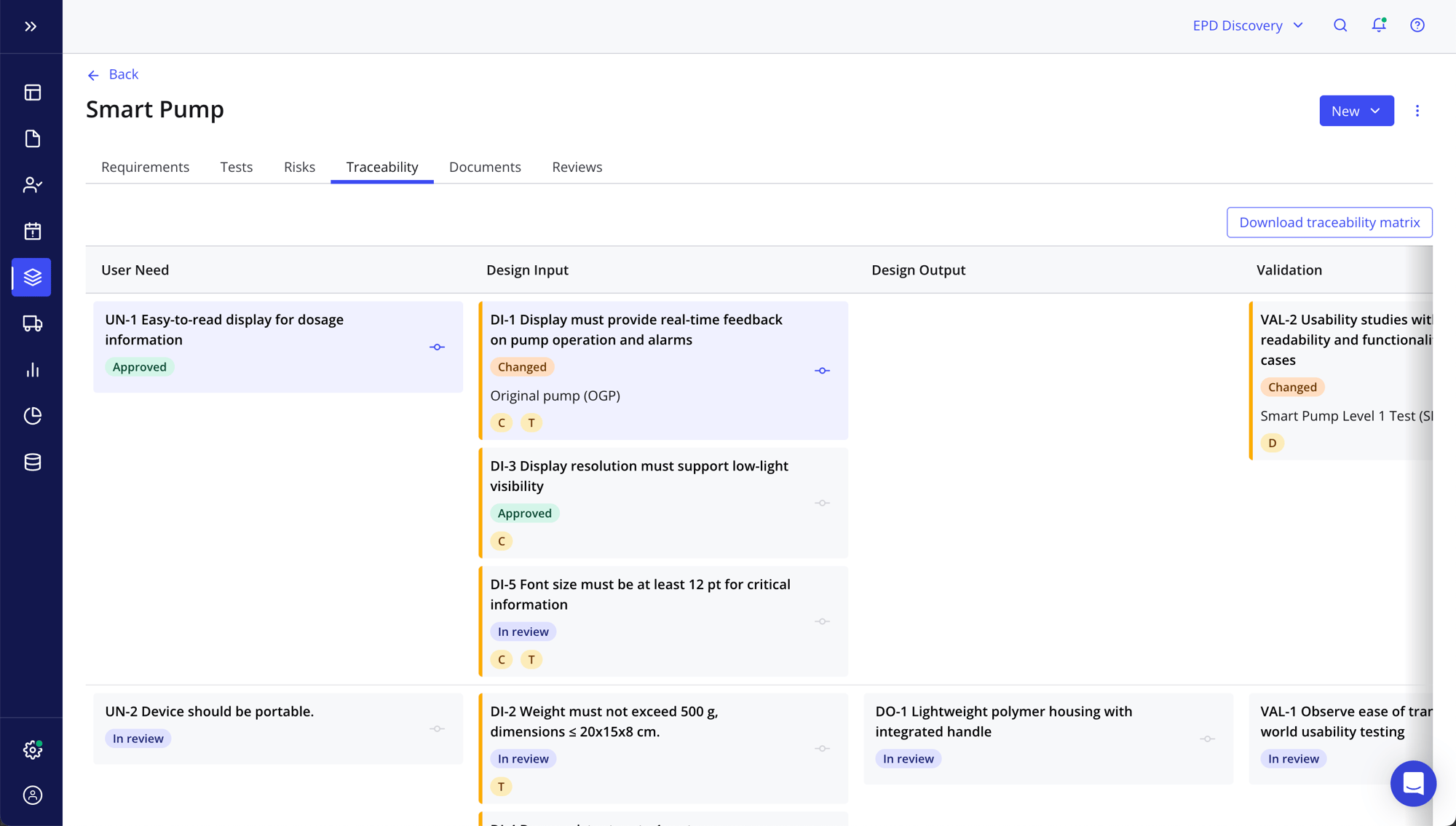
Generate release documentation with one click
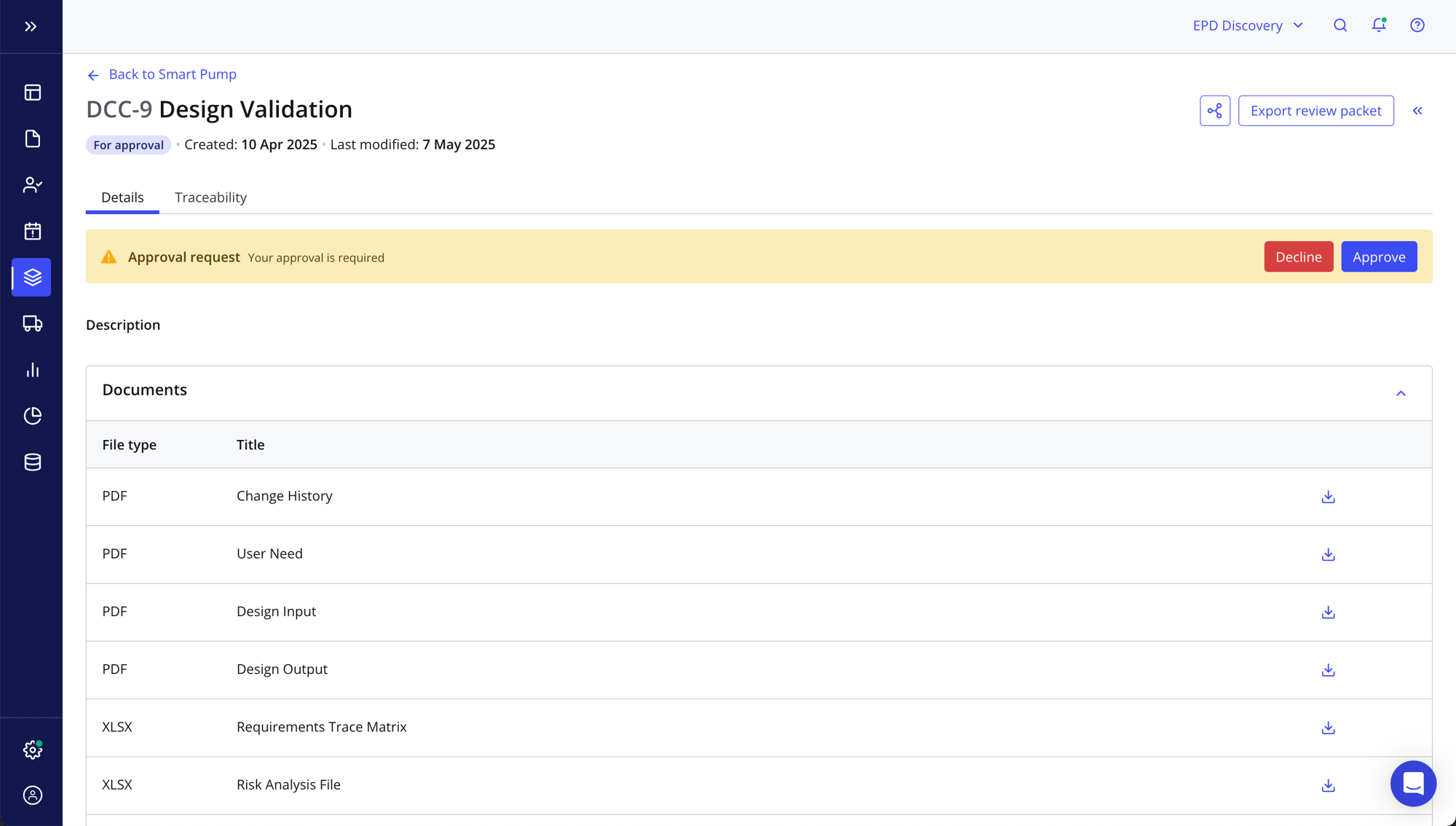
- Easily produce quality documentation to ensure you’re delivering a safe and effective product, eliminating time-consuming paper-based processes and manual governance
- Be 100% audit-ready at all times with documentation, e-signatures and objective evidence
- Access automated snapshots of product documentation, like a Change History Report, for each stage of your product lifecycle
Design control software purpose-built for your quality and product teams
Quality teams...
... for medical device, SaMD and SiMD companies enjoy a single source of truth for design controls and product data.
Export automated documentation, embed FMEA and ISO 14971 risk management and unlock complete traceability from input to output.
Product teams...
...enjoy uninterrupted development as the engineering systems they're used to are linked directly to audit-ready design controls management software.
Develop and innovate, safe in the knowledge that your work is flowing accurately and in real time straight to the quality department.
Design controls management resources
Ultimate guide to medical device design controls
Become a design controls expert and start applying the 9 core elements of medical device design controls with our comprehensive breakdown guide.

Design control software datasheet
Learn how our medical device design software combines powerful functionality with ease of use for a controlled, compliant pathway to market that meets ISO and FDA requirements.
See other features that make Qualio
the #1 top-rated cloud QMS
Document management
Make paper, filing cabinets, folders and spreadsheets a thing of the past.
Supplier management
Control and categorize suppliers and third parties with complete visibility.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Change control management
Manage change activity easily and effectively with complete traceability.
Audit management
Make compliance and audit readiness the natural core of your business DNA.