Map your QMS
Qualio consolidates all your SOPs, procedures, and objective evidence against QMSR requirements.
The FDA's new Quality Management System Regulation harmonizes 21 CFR Part 820 with ISO 13485. Qualio's AI-powered platform closes your gaps in weeks — not months.

The Quality Management System Regulation (QMSR), codified in 21 CFR Part 820, became effective on February 2, 2026. It harmonizes the FDA's device quality requirements with ISO 13485:2016, the international standard for medical device quality management systems.
Under QMSR, FDA now has authority to inspect management review records, internal audit reports, and supplier audit outputs.
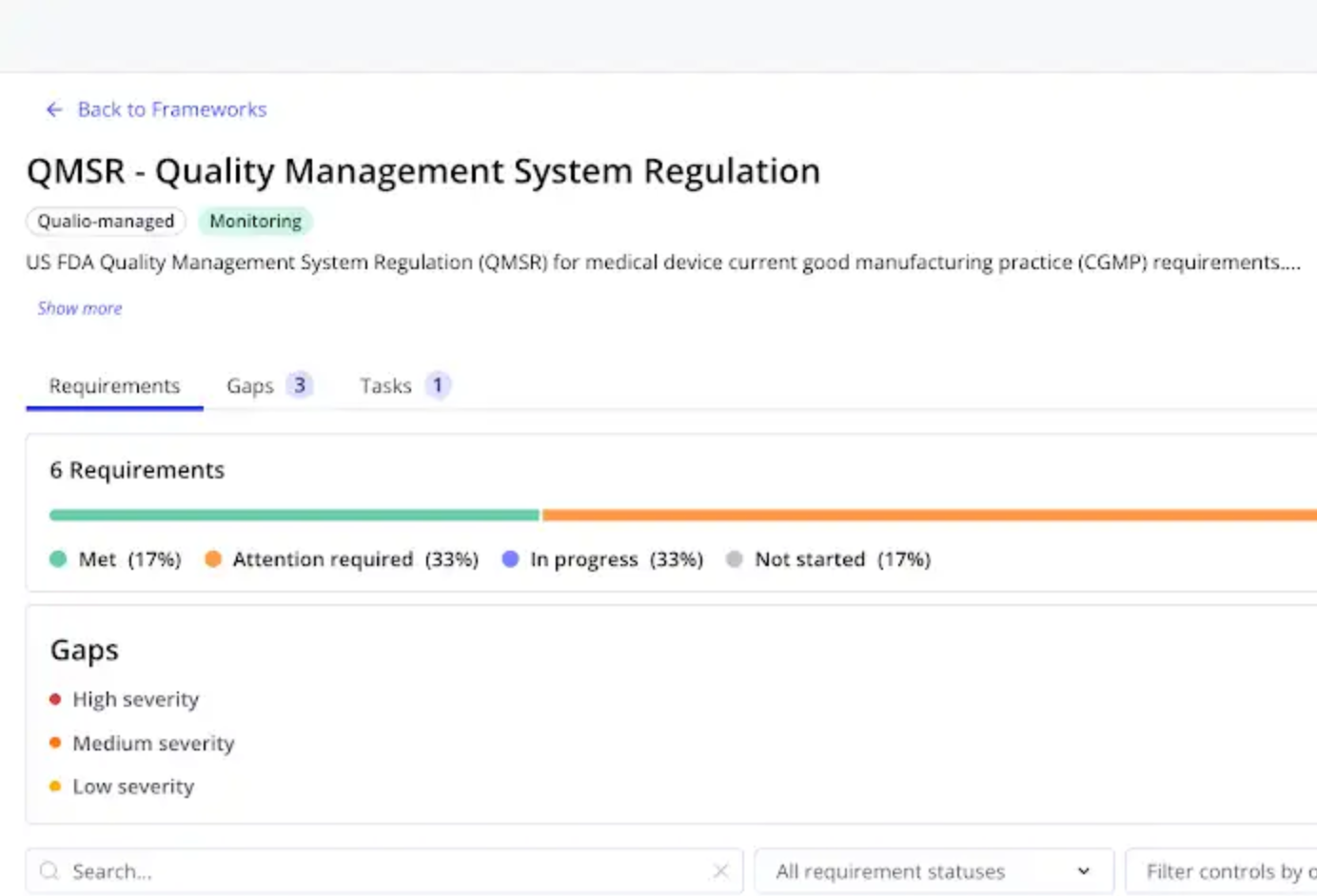
Qualio cross-maps your documents and processes against all QMSR clauses, surfacing gaps with evidence citations — not just checklists.
See your global compliance against QMSR and related requirements like ISO 13485 and MDSAP at a glance.
QMSR isn't a one-time project. Qualio monitors your compliance posture continuously — alerting your team to drift and gaps before they become observations.
No implementation sprints. No consultant dependency. Qualio's guided workflow takes you from connection to compliance — fast.
Qualio consolidates all your SOPs, procedures, and objective evidence against QMSR requirements.
Qualio generates a prioritized gap report and suggests remediation paths.
Assign owners, due dates, and get guided support on fixing gaps.
"Qualio gives us a 360° view of our QMS and automatically surfaces the gaps. We resolve issues faster, our team is more productive, and our risk is lower. With the system analyzing our documents and evidence end to end, we know we have full coverage and can walk into audits with total confidence."
When did the QMSR take effect?
February 2, 2026.
Does ISO 13485 certification mean I'm FDA-compliant under the QMSR?
No. ISO 13485 puts you in a strong position, but the QMSR includes FDA-specific requirements, particularly in 820.35 (record control) and 820.45 (labeling and packaging), that go beyond ISO 13485. You need to meet both.
Are FDA definitions still used under the QMSR?
Yes. FDA definitions for terms like "device," "labeling," and "combination products" take precedence over ISO definitions where there is any conflict.