ISO 14971 toolkit
Bringing a medical device to market? Airtight risk management is crucial.
Master ISO 14971 with our resource toolkit.
Instant access. No email required.
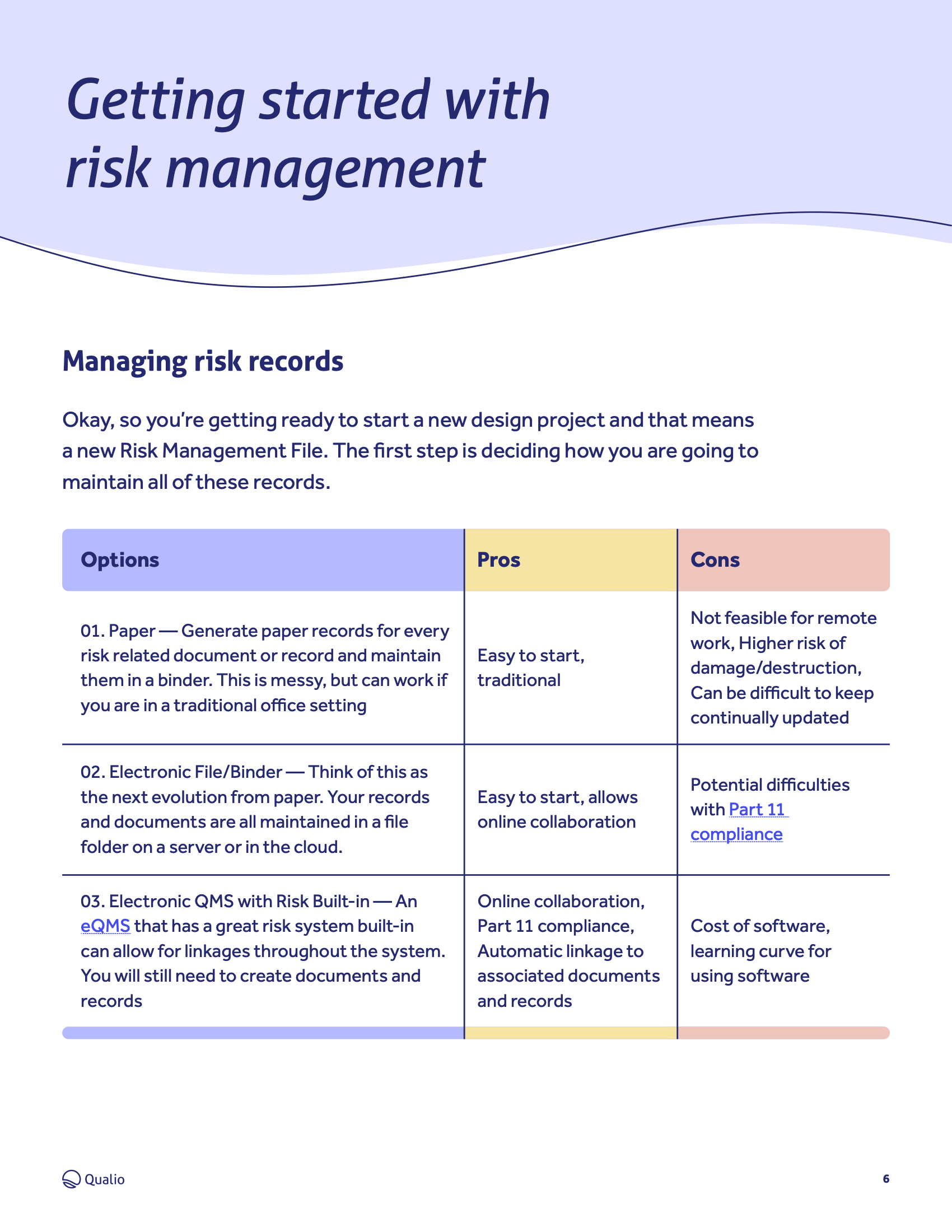
ISO 14971 is the industry risk management standard for organizations involved in the design, development and manufacture of medical devices.
Download our ISO 14971 toolkit PDF to:
- Get to grips with the requirements of the standard and what you need to do to comply
- Start taking actionable steps towards robust and long-term ISO 14971 compliance
- Become completely audit-ready and bring a safe, high-quality and risk-controlled medical device to market
What you'll get:
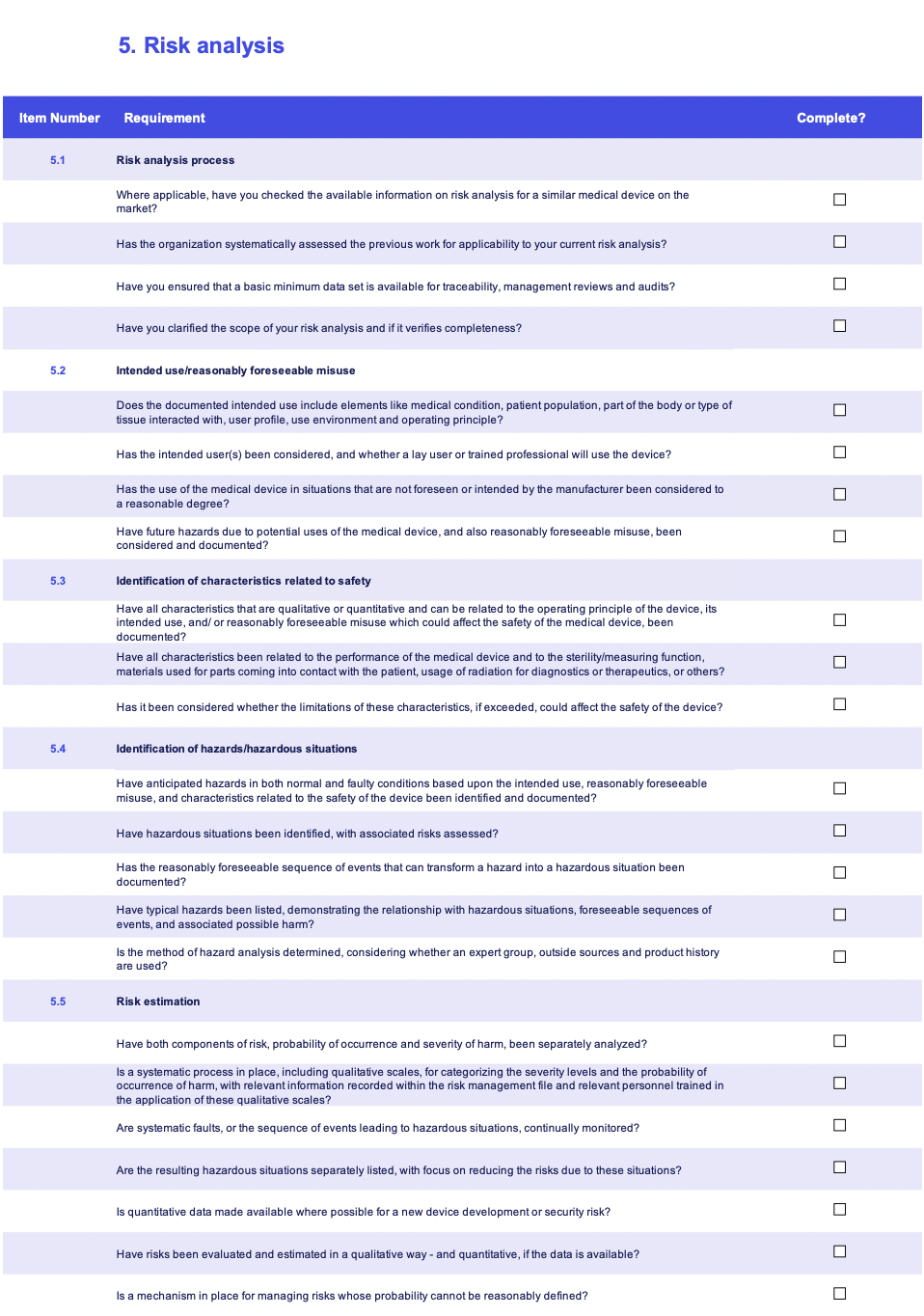
Compliance checklist
Introduction to medical device risk management
ISO 14971 mastery guide
Master the requirements of medical device risk management, from risk analysis to assembling your RMF
Mastering risks and opportunities guide
What can Star Wars teach us about risk? Explore the broader principles of risk management with some help from Yoda, Luke Skywalker and even Darth Vader