PHARMA GxP COMPLIANCE
GxP compliance for pharma teams
Create life-saving drugs with a purpose-built pharmaceutical compliance platform.
Qualio streamlines your quality processes while keeping your teams compliant with
FDA, ISO, EMA and GxP regulations.

Take the tour
QMS software for the pharmaceutical industry
Unlock simpler compliance with our pharma QMS software
Challenge
Pharmaceutical companies work in a high-pressure, highly regulated environment where the consequences of errors can be severe.
You have to balance regulatory compliance, quality assurance and the ethical responsibility of ensuring that the medications your patients rely on are safe and effective.
And with the pace of innovation accelerating, the traditional paper-based QMS for pharma operations begins to creak under the weight.
Solution
Qualio's pharmaceutical QMS software makes it easy to manage your quality demands, from FDA, EMA and ICH compliance to GxP adherence.
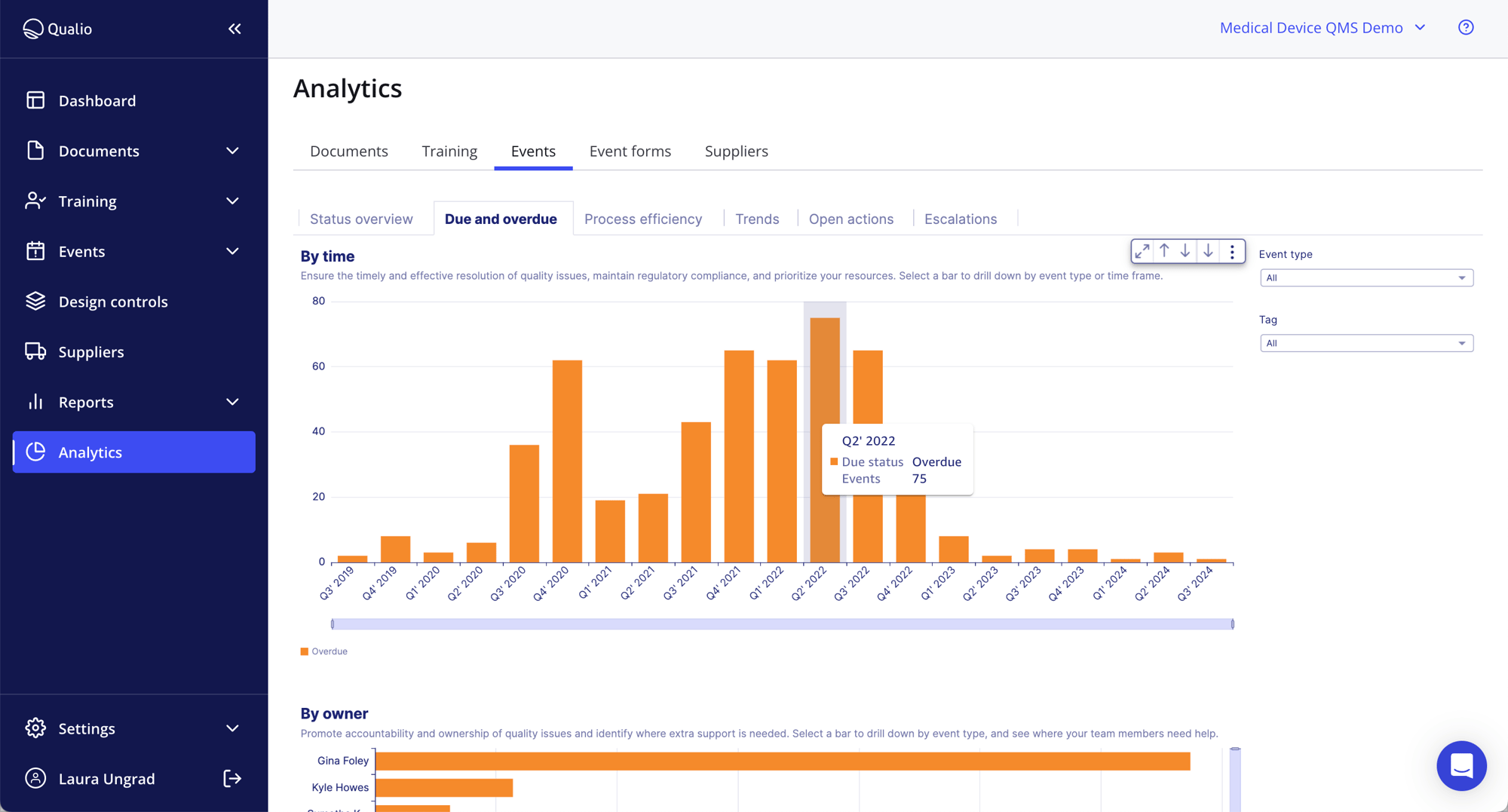
With flexible workflows and templates tailored to your needs, our pharma eQMS scales with you. Easily maintain detailed records and documentation for every aspect of your operation, underpinned by detailed reporting on training, change control, suppliers, events, audit trails and more.
An end-to-end pharma QMS solution from development to production
- Embed quality with an all-in-one digital pharma QMS platform that scales as you develop new drugs, departments and suppliers
- Unite your team and integrate with your business-critical apps for effective collaboration and maintain data integrity for quality assurance
- Create and execute workflows for document approval, assignment of tasks, training and more
- Manage and report on suppliers, audits and change control across your entire business in real time
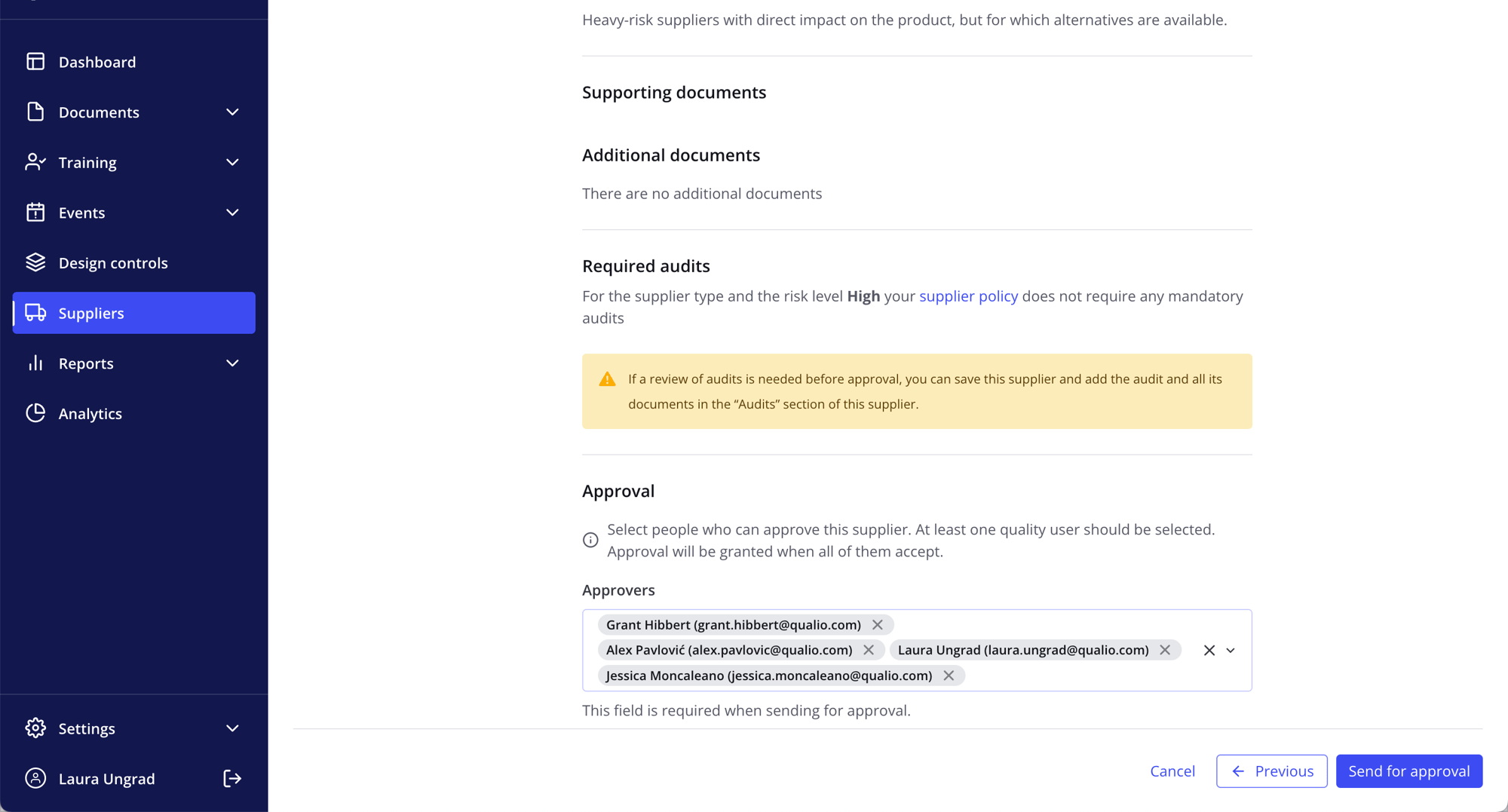
Developing medicinal product is hard! (Using Qualio isn’t.)
- Our pharmaceutical QMS software is ranked the easiest to set up on G2 - so onboarding is easy for your team members
- Effortlessly migrate your legacy pharma QMS. Digitize existing documentation and quickly generate release documentation with a single click
- Seamlessly collaborate on documents and events with our in-app editor
- Strengthen pharmacovigilance with easy and effective event creation, management and reporting
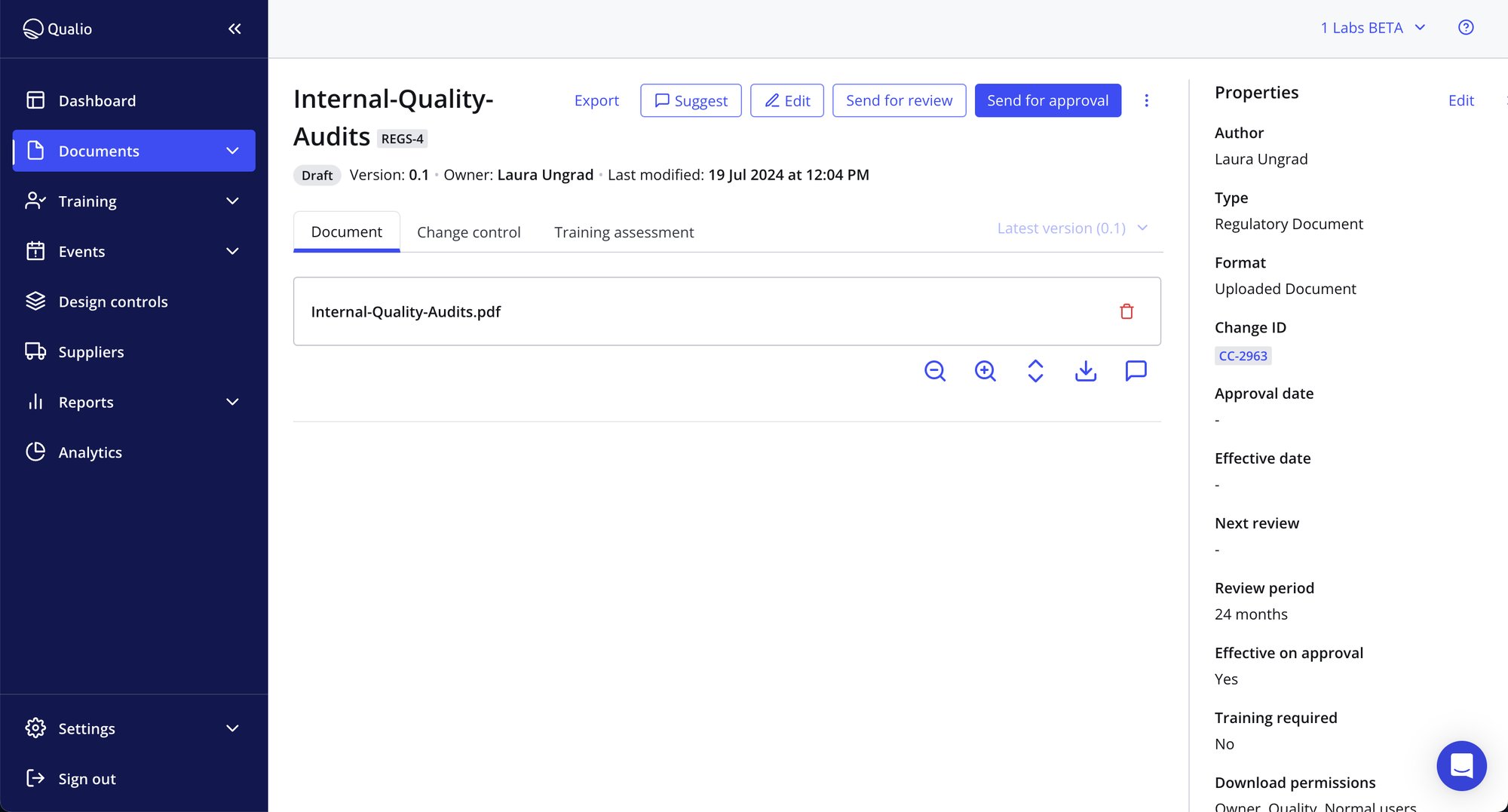
Comply with all your standards and be constantly audit-ready
- Easily generate detailed reporting on training, overdue and completed activities, audit trails, change control, event and supplier management and more
- Set periodic review cadences to embed GxP and meet FDA, ICH, EMA and ISO requirements
- Assign CAPA, deviation and OOS tasks to your team and track through to completion
- Accelerate your route to market with support from our in-house quality experts
Pharmaceutical quality management resources
Pharmaceutical quality software datasheet
Learn how Qualio provides a holistic and integrated PQS for your business.

Pharmaceutical quality management software guide
Read our blog post to dive deeper into pharmaceutical quality software and how to choose the best system for you

ICH Q10 toolkit
Access a range of resources to kickstart your ICH Q10 compliance journey, from a step-by-step checklist to a detailed preparation guide.
Don't just take our word for it!
Hear from our happy pharma QMS software customers
“My favorite thing is how Qualio keeps us in a constant state of audit readiness.”
Deb G.
Director of Quality, Dimension Therapeutics
"The Qualio team made a big difference to our company by providing amazing help and guidance at a critical time in our QMS development."
Jackie L.
Managing Director, Auxilio Pharmaceuticals
"Qualio is the best QMS cloud-based system I have ever seen."
Paul H.
Senior VP of Ops, Tech & Quality, Advantice Health
"The perfect QMS system for a start-up. Everything, from validation to migration and training, was a positive experience."
Drew M.
Director of Quality, ECM Therapeutics
"A game-changer for a paper-heavy QMS. Qualio is a breath of fresh air."
Gareth Q.
Director of Quality, iMIX
"With other vendors, we felt like just a number.
But this is like a true partnership. Qualio has definitely changed our world."
Eric D.
Director of Quality Assurance, Expression Therapeutics
More content for pharmaceutical
professionals

From Lab to Launch
Hear from real pharmaceutical scientists, engineers, innovators and business leaders. New episodes every week!

GxP toolkit
Access a range of helpful GxP resources, including a compliance checklist, GAMP 5 guidance and more.
What your QMS needs for ISO and FDA regulation
Your pharmaceutical QMS needs to make your FDA and ISO auditors happy. Find out how with our webinar recording.
