AUDIT MANAGEMENT
Always audit-ready, not just audit-prepped
Make stressful, last-minute audit prep a thing of the past. Continuous readiness across every framework, with evidence collected automatically.

Audit management software for smarter, calmer audits
Challenge
Internal audits can be burdensome, resource-heavy slogs.
External audits can be stressful nightmares upon which the future of your business rests.
And failing to prepare can lead to fines, penalties or product recalls.
Solution
Qualio gives your team the audit management tool you need for maximum, constant audit readiness.
Access data, prove compliance, demonstrate improvement and respond to findings. Make internal and external audits quick, enjoyable growth drivers - not nail-biting tests.
Be 100% compliant and audit-ready
- Identify gaps and optimize quality performance with a connected audit management system
- Have confidence your QMS is fully compliant with ICH Q10, ISO 9001, ISO 13485, ISO 14971, FDA requirements, and the EU MDR (to name a few!)
- Prove continuous improvement from past audits with organized documents, procedures and automated internal QMS audit schedules and tasks
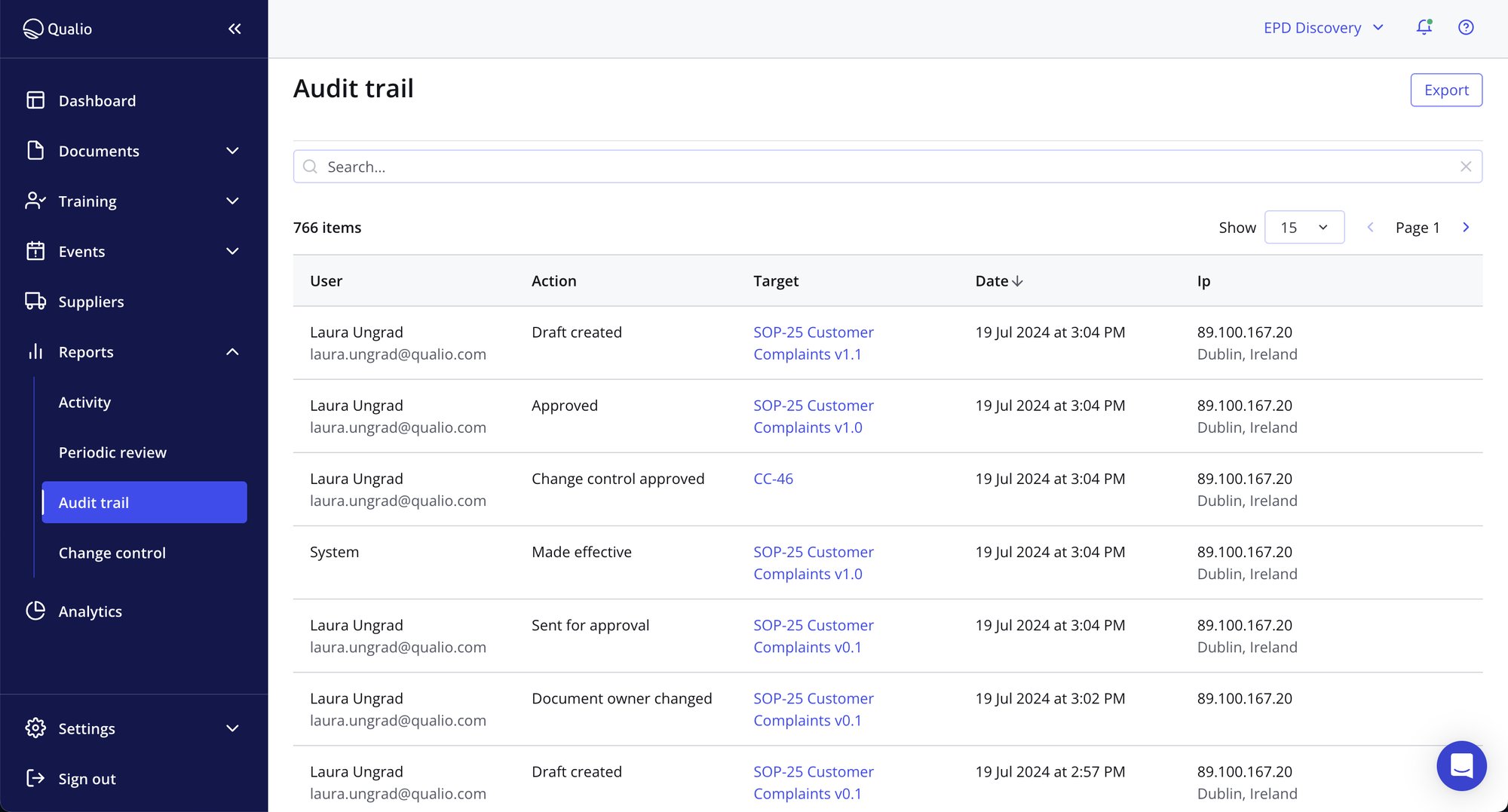
Save time with automated document and data processes
- Ditch the challenges of paper-based audits with cloud-based quality audit software that makes remote audits simple
- Find all audit artifacts with one click using document templates and tagging
- Build bespoke workflows to assign tasks and automate reminders from one place
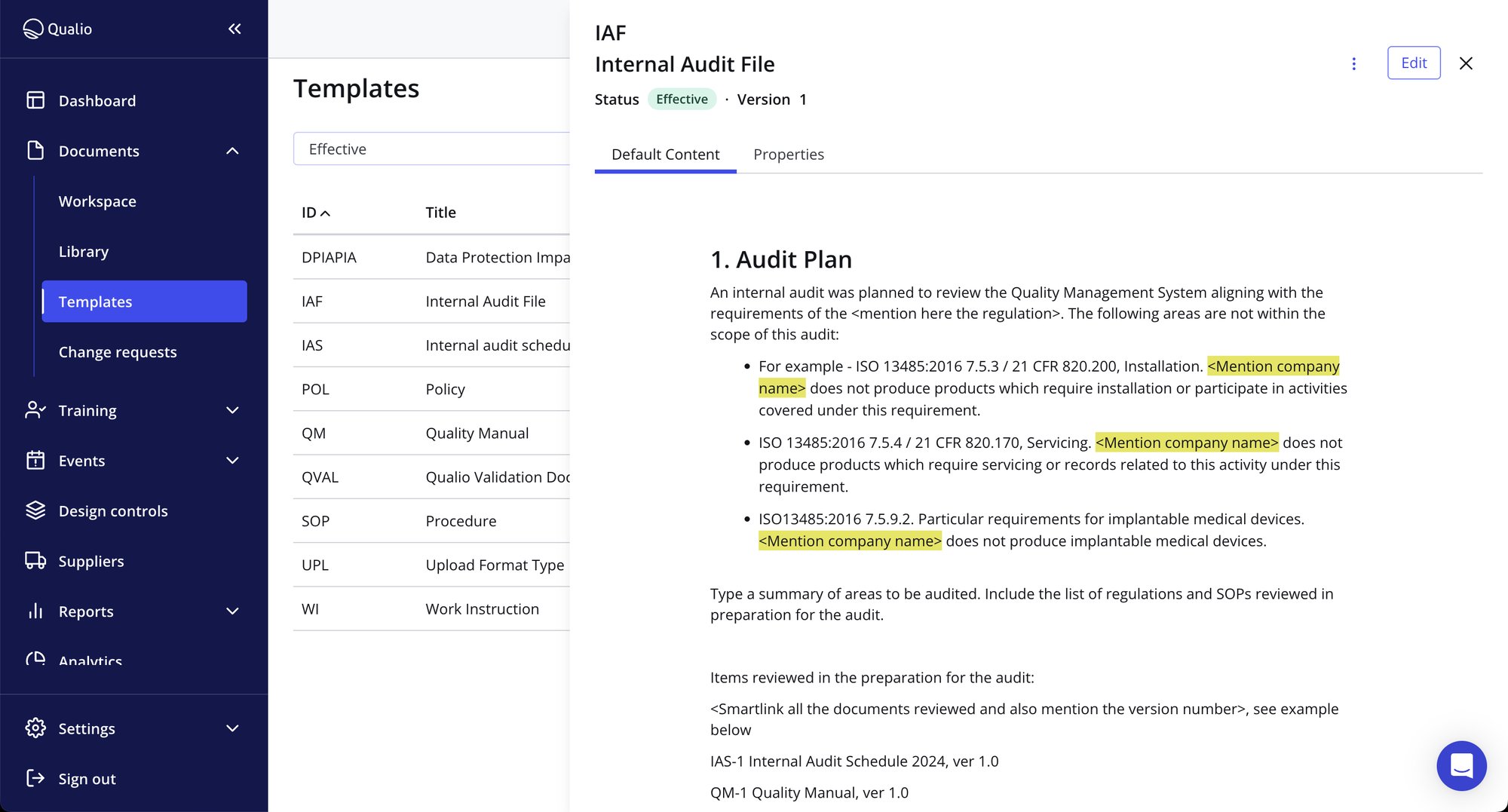
Make audit readiness your competitive advantage
- Squash the risk of fines, penalties and product recalls by connecting your audit management program, data and teams to a centralized audit management software platform
- Easily assign QMS audit tasks, manage due dates and track findings to completion - all from a single audit management solution
- Enjoy end-to-end traceability for all CAPAs, non-conformances, and other quality events to demonstrate complete control and visibility to your auditors
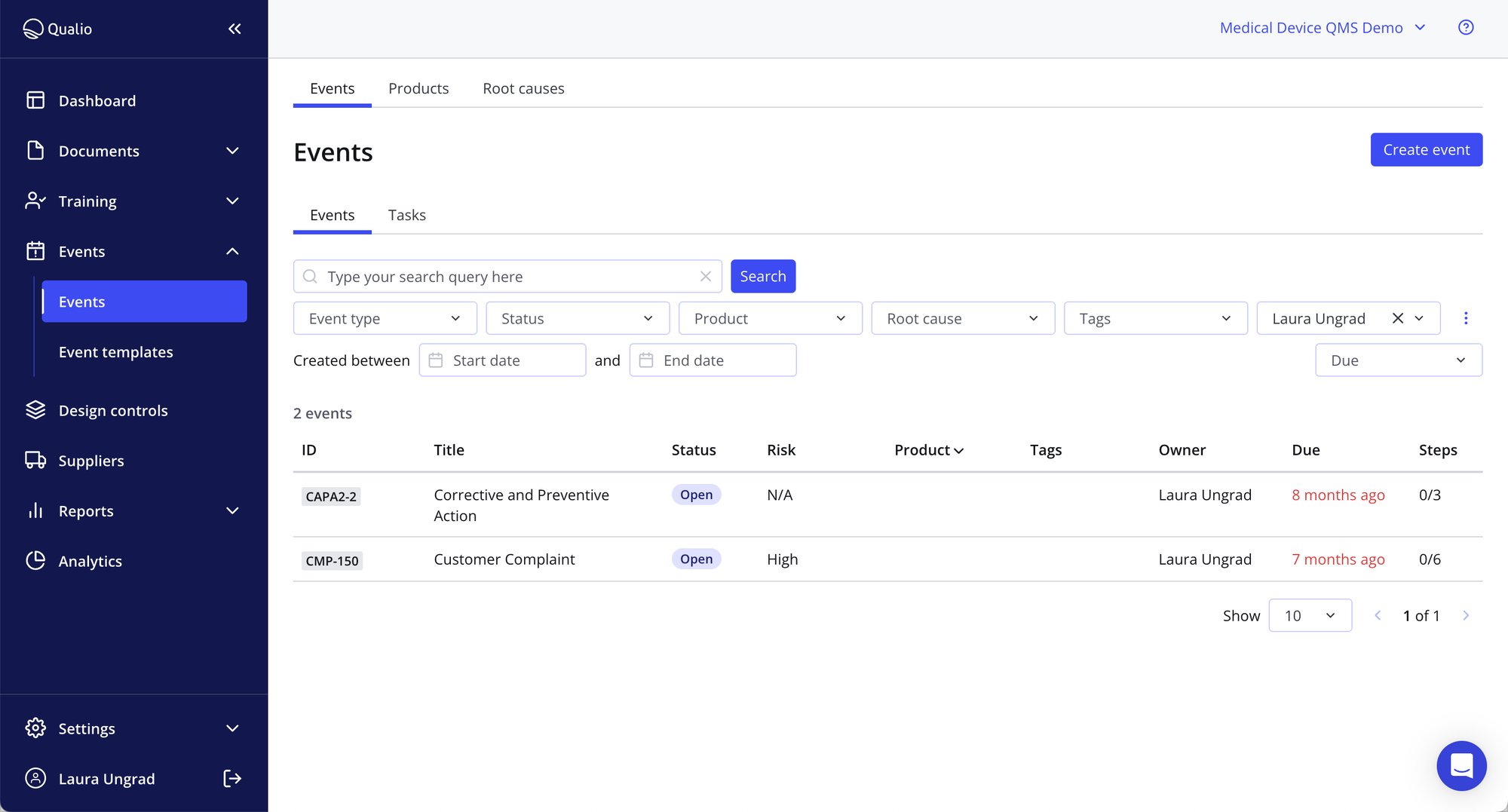
Quality management audit resources

Audit readiness checklist
Pinpoint gaps, take targeted action and get your QMS ready for your next audit.

Passing an audit webinar
Hear tips, insight and best practice from a webinar panel of seasoned quality experts.
See other features that make Qualio
the #1 top-rated audit compliance software
Supplier management
Control and categorize suppliers and third parties with complete visibility.
Document management
Make paper, filing cabinets, folders and spreadsheets a thing of the past.
Design control management
Supercharge your product development from a single source of truth.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Change control management
Manage change activity easily and effectively with complete traceability.