TRAINING MANAGEMENT
Record changes. Training follows.
When a record changes, affected training assignments update automatically. Assign courses, track completions, and generate audit-ready training records. No spreadsheets.

GxP training compliance without the manual overhead
Challenge
Many quality teams today struggle to know whether their colleagues are up-to-date on the latest procedures and QMS training requirements.
The training management challenge gets harder with disparate systems, in a remote environment, or with paper. Quality managers run around to gather training signatures without complete compliance confidence.
Solution
With Qualio’s cloud-based training management software, you can assign training based on roles and responsibilities, including in-line assignments.
Build training workflows for newly approved documents, send reminders automatically, and generate reports showing training status to ensure you stay compliant.
Centralize e-learning content in one training management software system
- Make Qualio your one-stop shop for QMS training, creating efficiency across your company and lowering risks of losing important knowledge
- Make training management easier with streamlined, role-based quality management system training
- Simplify compliance and reduce staff educational costs through consolidation into a single training management system software platform
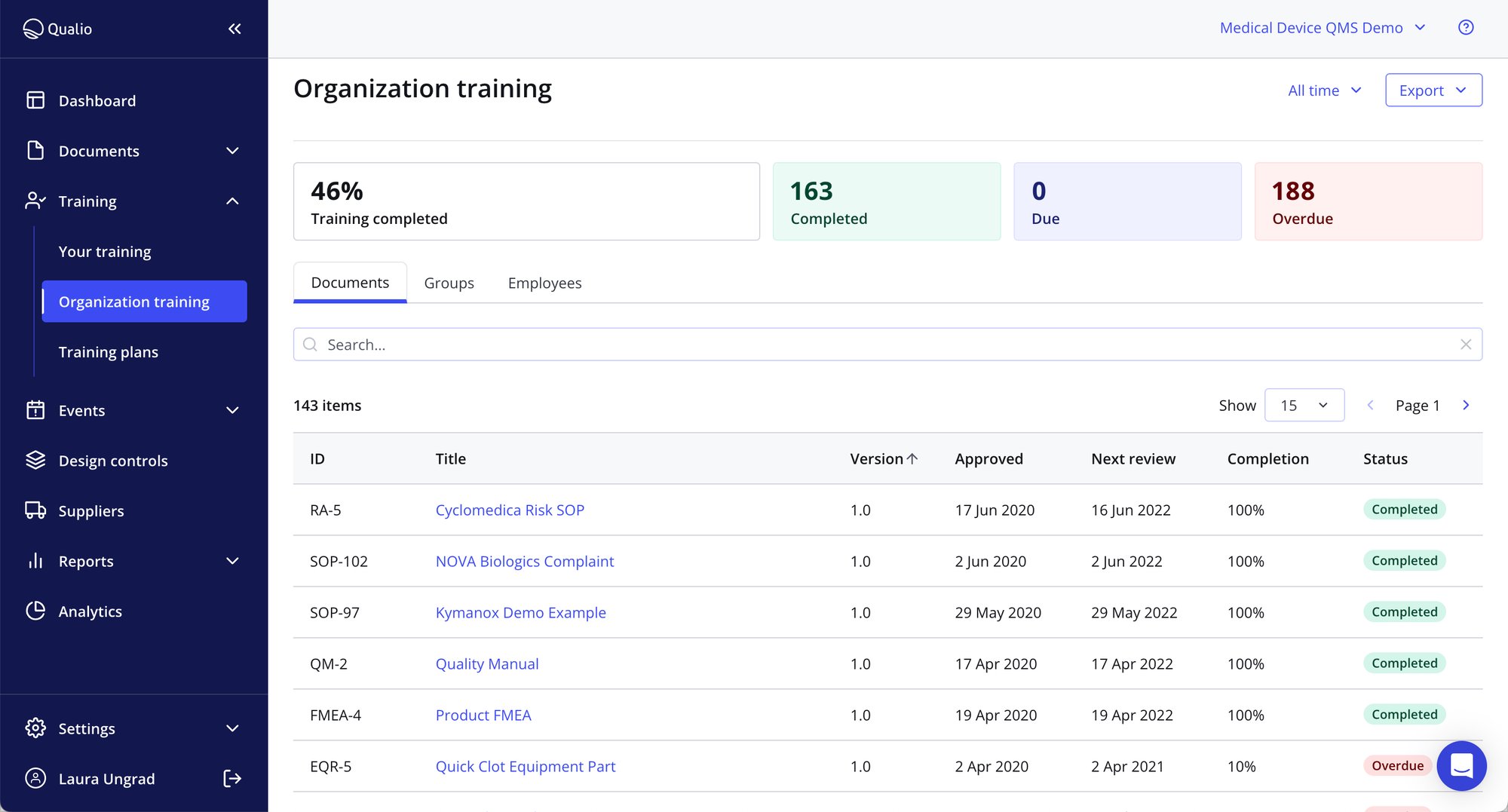
Personalized training based on role and responsibilities
- Assign the right training to the right person including in-line assignments
- Build training software workflows so only those who need training on new procedures get notified
- Streamline training to a team member in response to quality events or CAPA
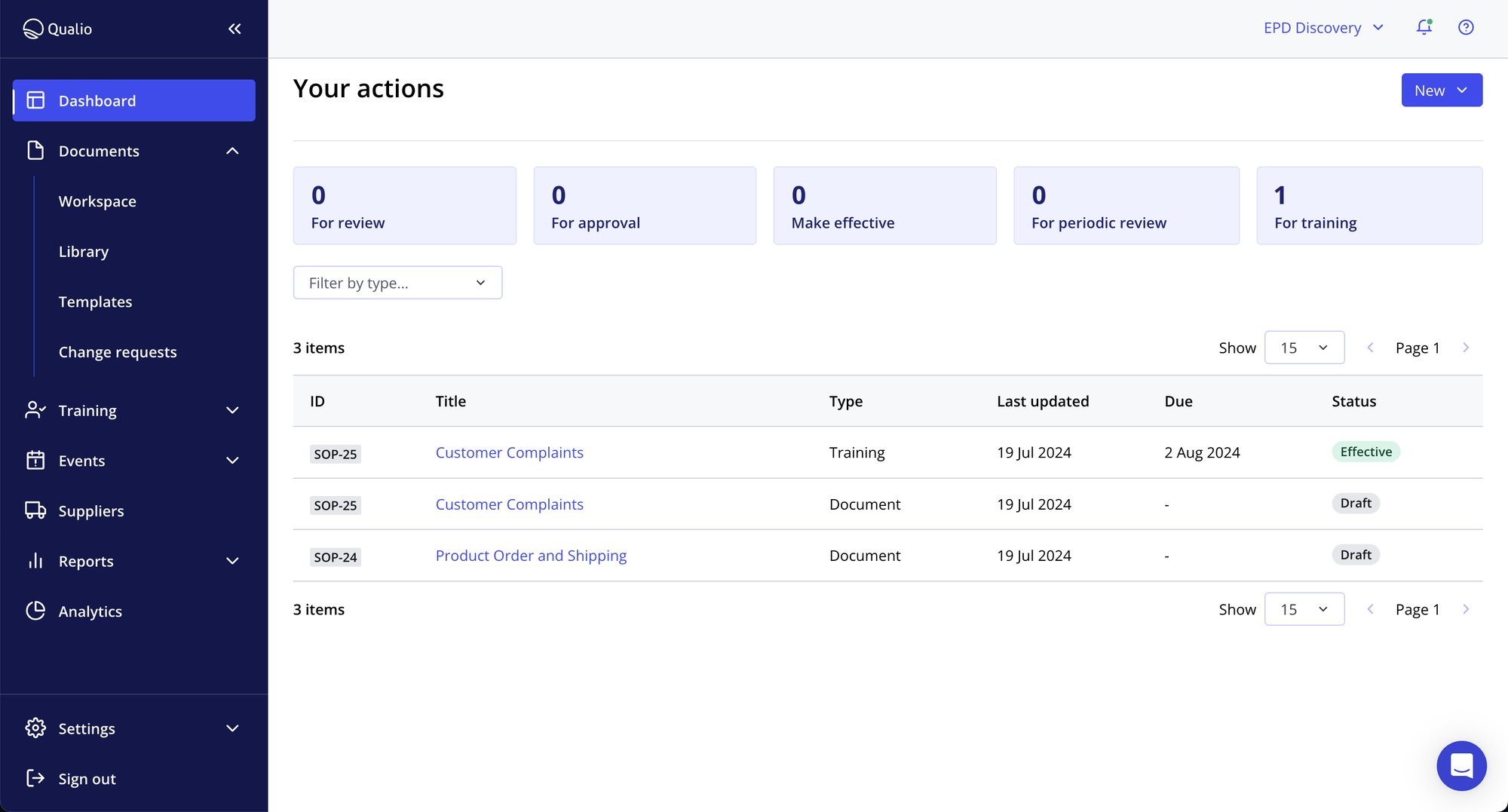
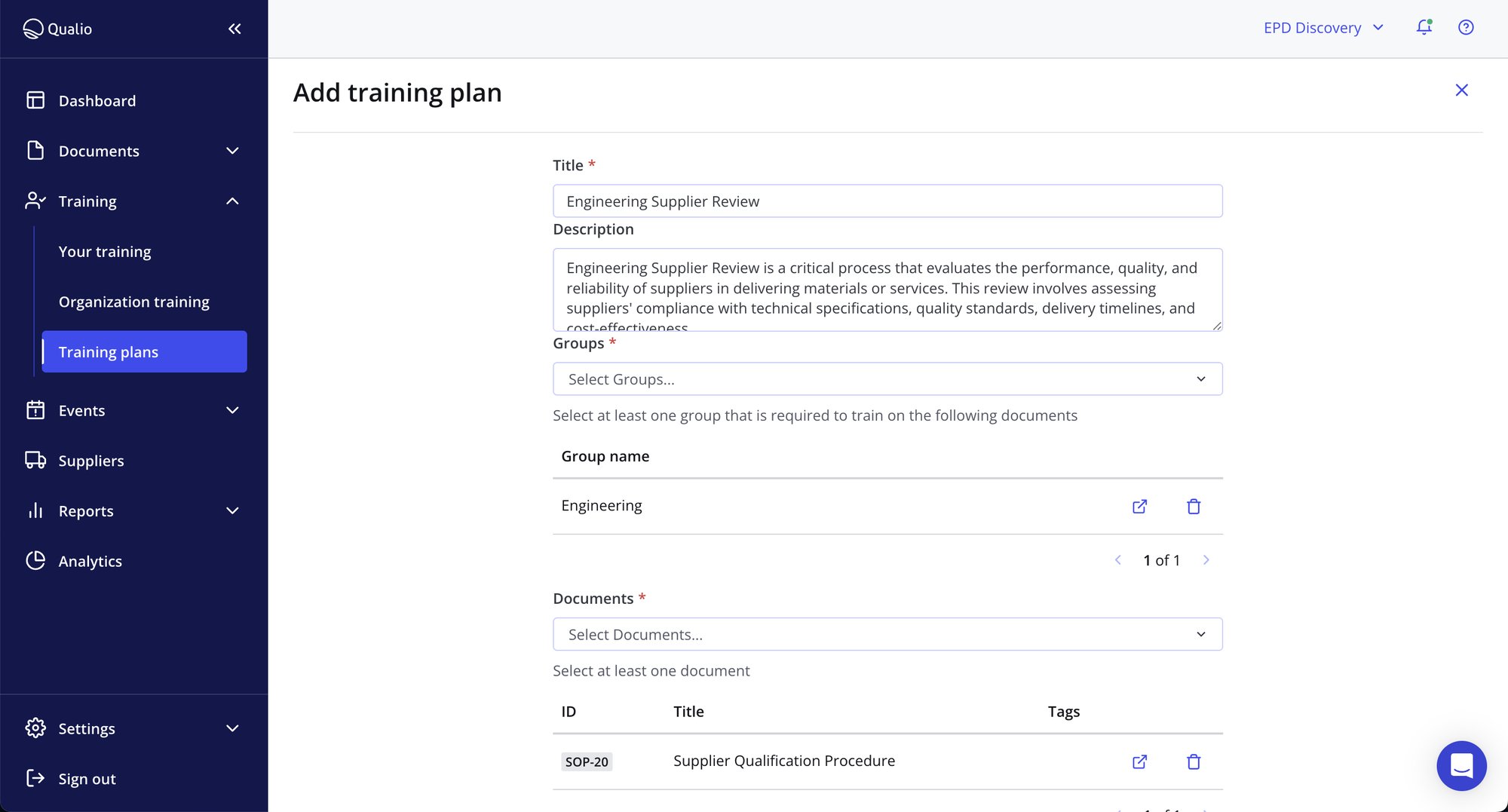
Cascade training business-wide with Training Plans
- Use Training Plans to train groups, teams and departments at the touch of a button
- Bundle training requirements and push to system users and groups quickly and easily
- Parcel training tasks by seniority, start date, ISO standards and more
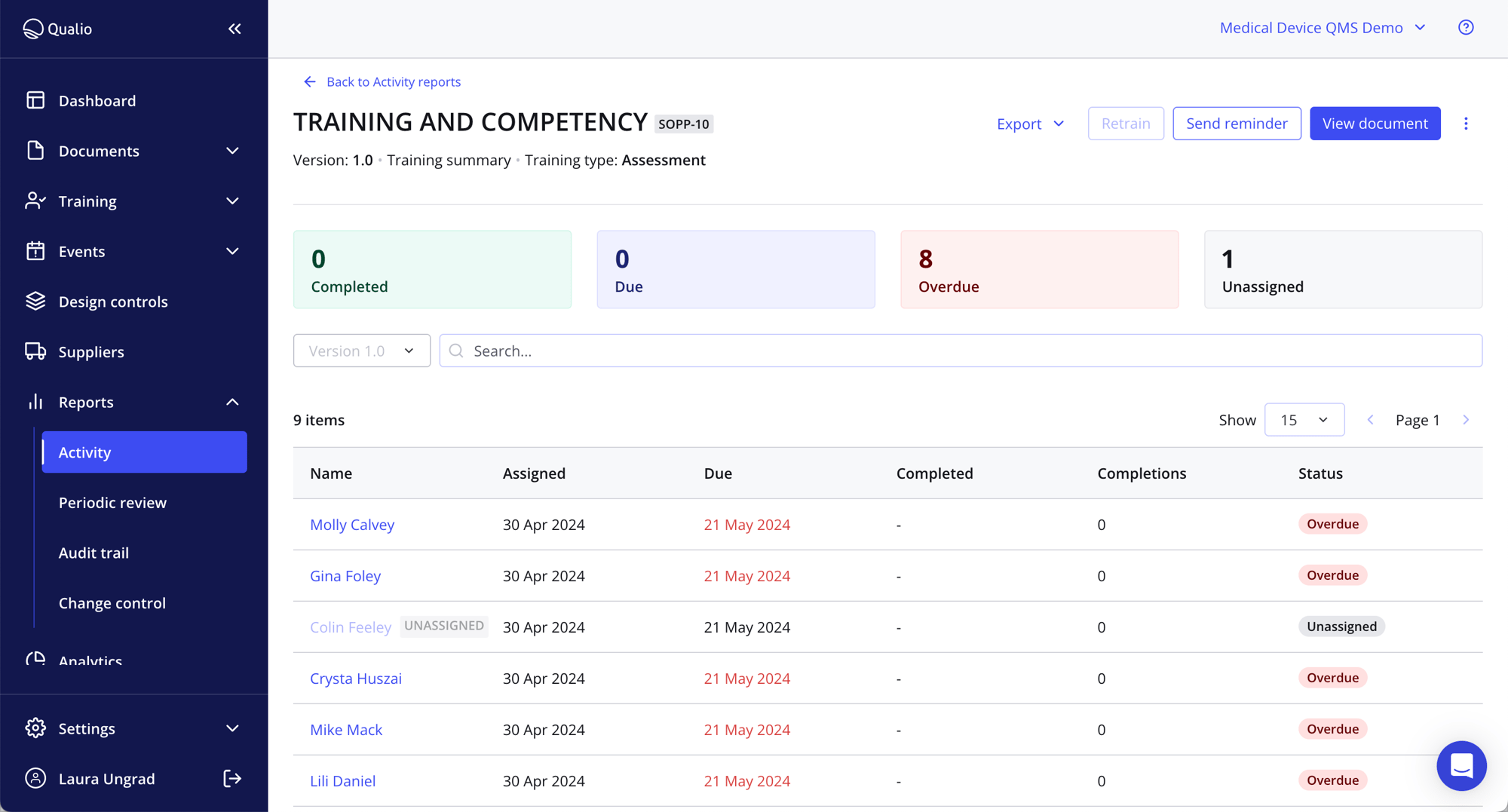
Ensure compliance and competency by tracking completion
- Cut the stress with training compliance software underpinned by Part 11- and Annex 11-compliant e-signatures
- Report on QMS training to identify and mitigate gaps, ensuring audit readiness
- Create quality system training assessments to maintain quality standards and compliance
Quality management system training resources

Training management software datasheet
Dive into our training management system software and the powerful operational benefits you can expect.

Culture of quality
toolkit
Download our toolkit and learn how to train, educate and empower your colleagues towards a real culture of quality.
See what else Qualio does for training
Document management
Make paper, filing cabinets, folders and spreadsheets a thing of the past.
Supplier management
Control and categorize suppliers and third parties with complete visibility.
Design control management
Supercharge your product development from a single source of truth.
Change control management
Manage change activity easily and effectively with complete traceability.
Audit management
Make compliance and audit readiness the natural core of your business DNA.