RISK MANAGEMENT
Identify and mitigate risks before they become findings
Proactive risk identification, mitigation, and resolution in a dedicated risk platform. Connected to your design controls and quality events.

Risk management software solutions to give your business confidence
Challenge
Risks lurk in every nook and cranny of your business. Pinpointing and tackling every single one is extremely difficult.
And when you do get a risk management system in place, it doesn't always integrate with your product design and development processes - leaving crucial risks undetected and unresolved as you get to market.
Solution
Qualio's risk software embeds risk management into the heart of your product design and development activities.
Total visibility, bespoke action workflows and automated document stack creation removes the effort and headache of risk management and lets you bring a safe, compliant product to your patients and customers.
Detect vulnerabilities across your entire organization
- Build a single source of truth for risk identification, monitoring and remediation
- Save time and money by identifying risks early in product development
- Connect your quality and product teams in a single risk assessment software environment that keeps risk-based thinking front and center
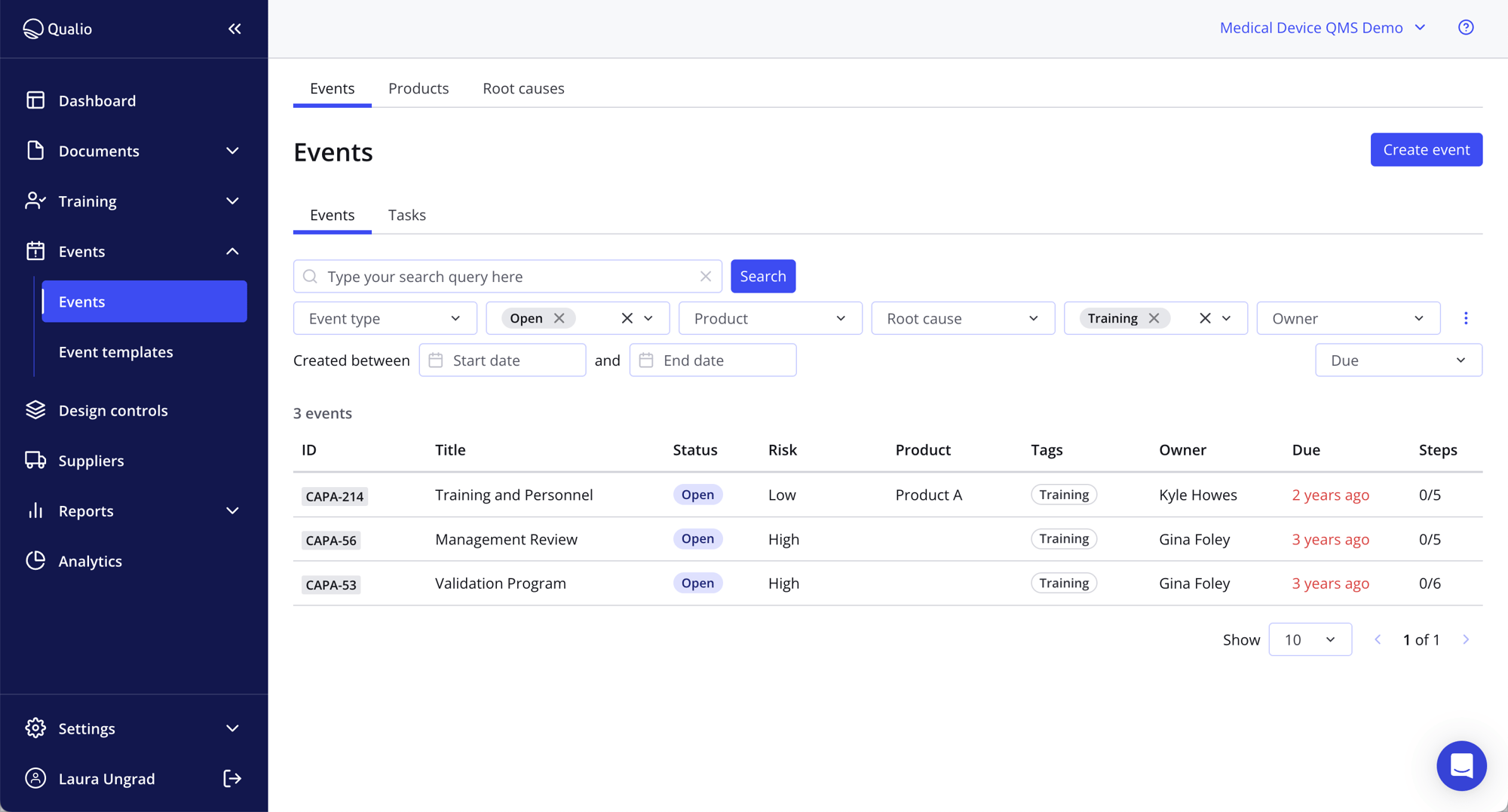
Unlock deep risk visibility with an acceptability matrix
- Define your risk criteria then apply them across a connected risk management software platform
- Link risks to your product development design control and documentation processes to squash risks for a right-first-time approach
- Seamlessly access risk analysis and traceability matrices with one click
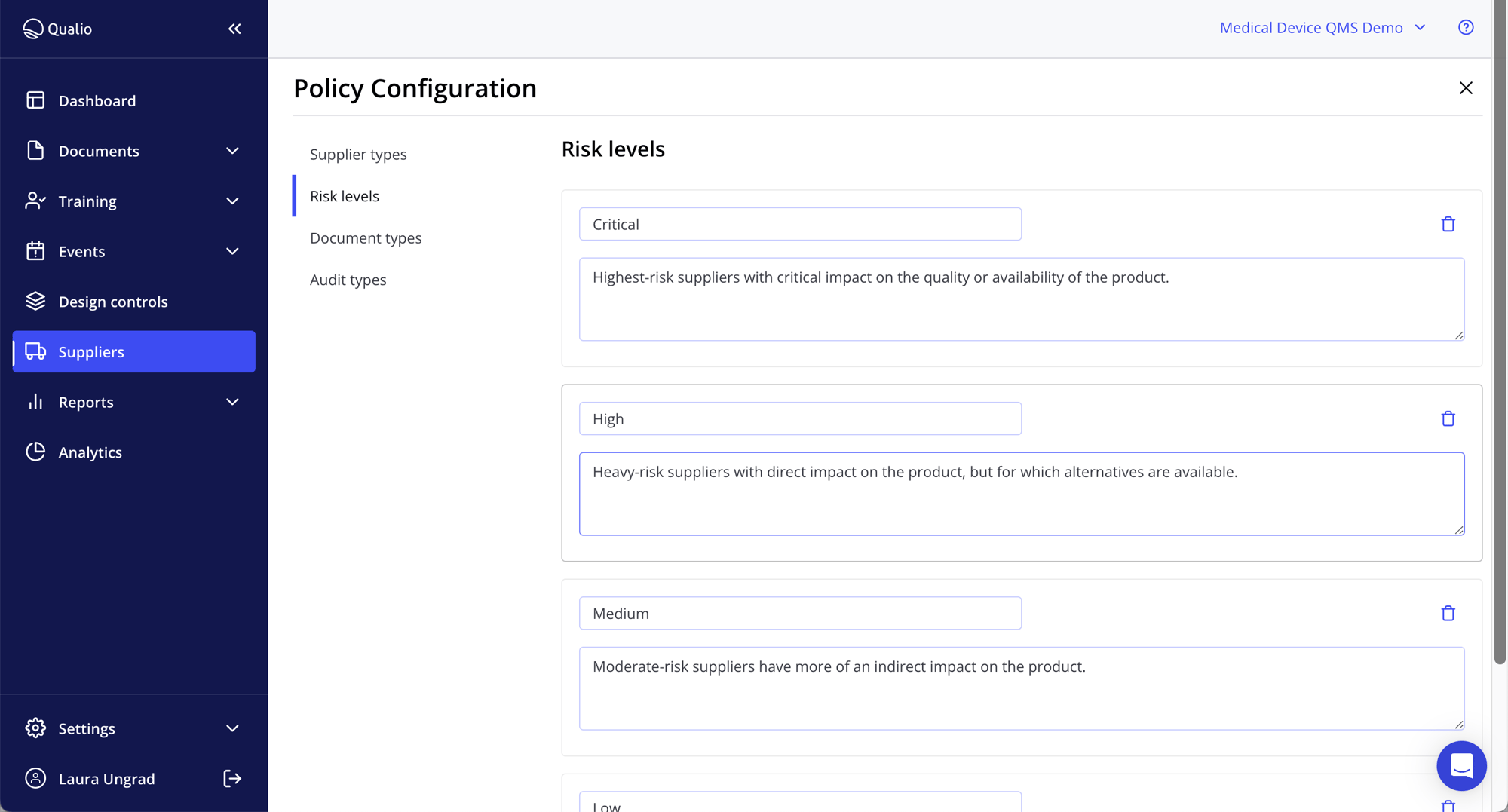
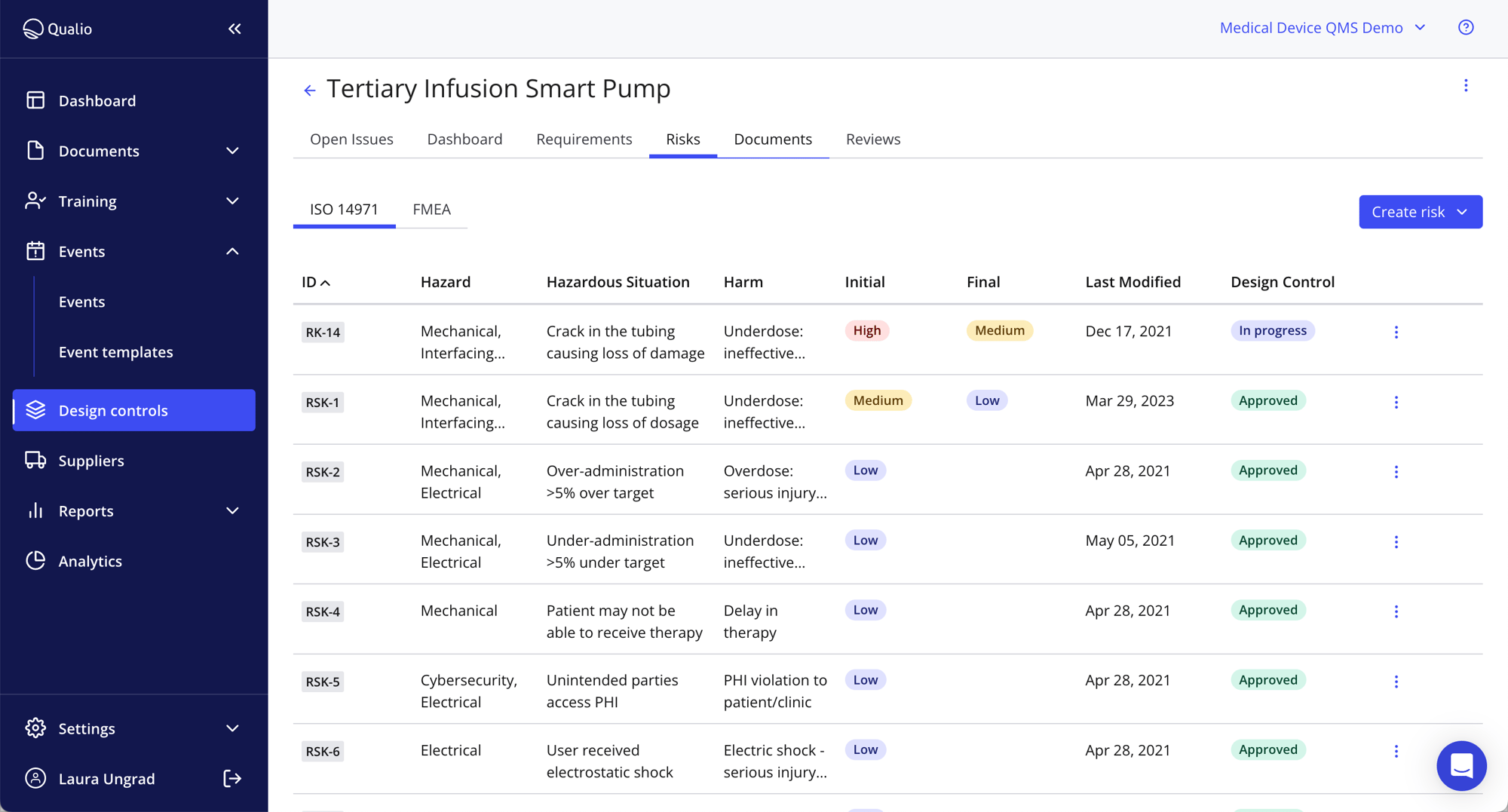
Embed ISO 14971 and FMEA
- Use our ISO 14971 software to embed lasting compliance into your medical device design controls
- Show complete FMEA-based risk control to your auditors
- Export risk records and link risks to CAPA plans and Qualio documents
Quality risk management system resources

ISO 13485 risk management plan template
Build a structured medical device risk management plan with our pre-built template.

Mastering risks and opportunities webinar
Build a holistic risk management system that squashes risk and harnesses opportunity.
See other features that make Qualio
the #1 top-rated compliance and risk management software
Document management
Make paper, filing cabinets, folders and spreadsheets a thing of the past.
Design control management
Supercharge your product development from a single source of truth.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Change control management
Manage change activity easily and effectively with complete traceability.
Audit management
Make compliance and audit readiness the natural core of your business DNA.
Supplier management
Control and categorize suppliers and third parties with complete visibility.