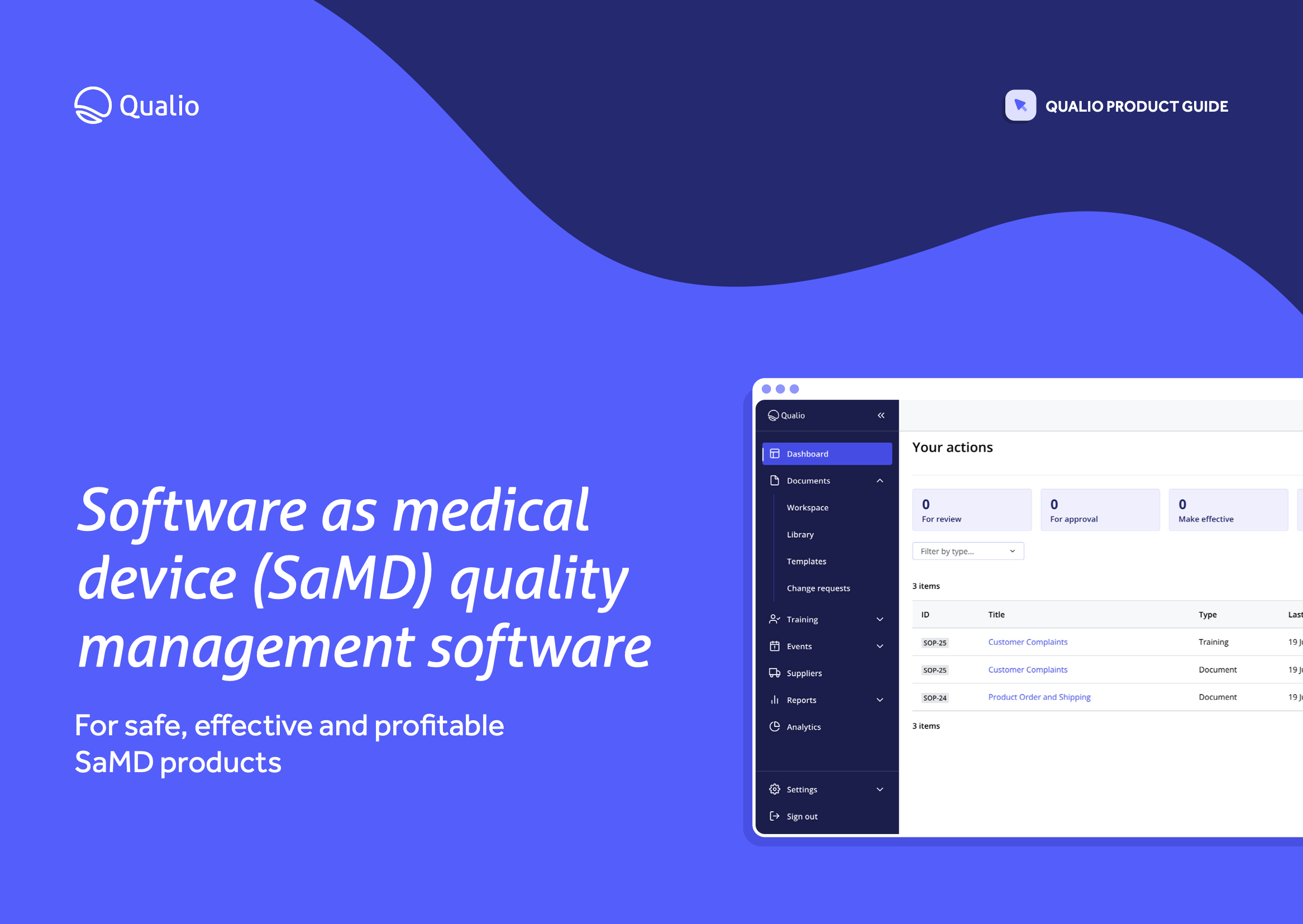
SaMD quality management software datasheet
Build a best-in-class, fully digital SaMD quality system.
Download our SaMD quality management datasheet to learn how Qualio:
- Arms your business with a complete, compliant eQMS for software as medical device quality management
- Supports and simplifies compliance with the demands of IEC 62304, ISO 13485, ISO 14971, the EU MDR, and more
- Digitizes and automates all areas of SaMD quality management, from design controls to document management
Complete the form to the right to get started!
What you'll get
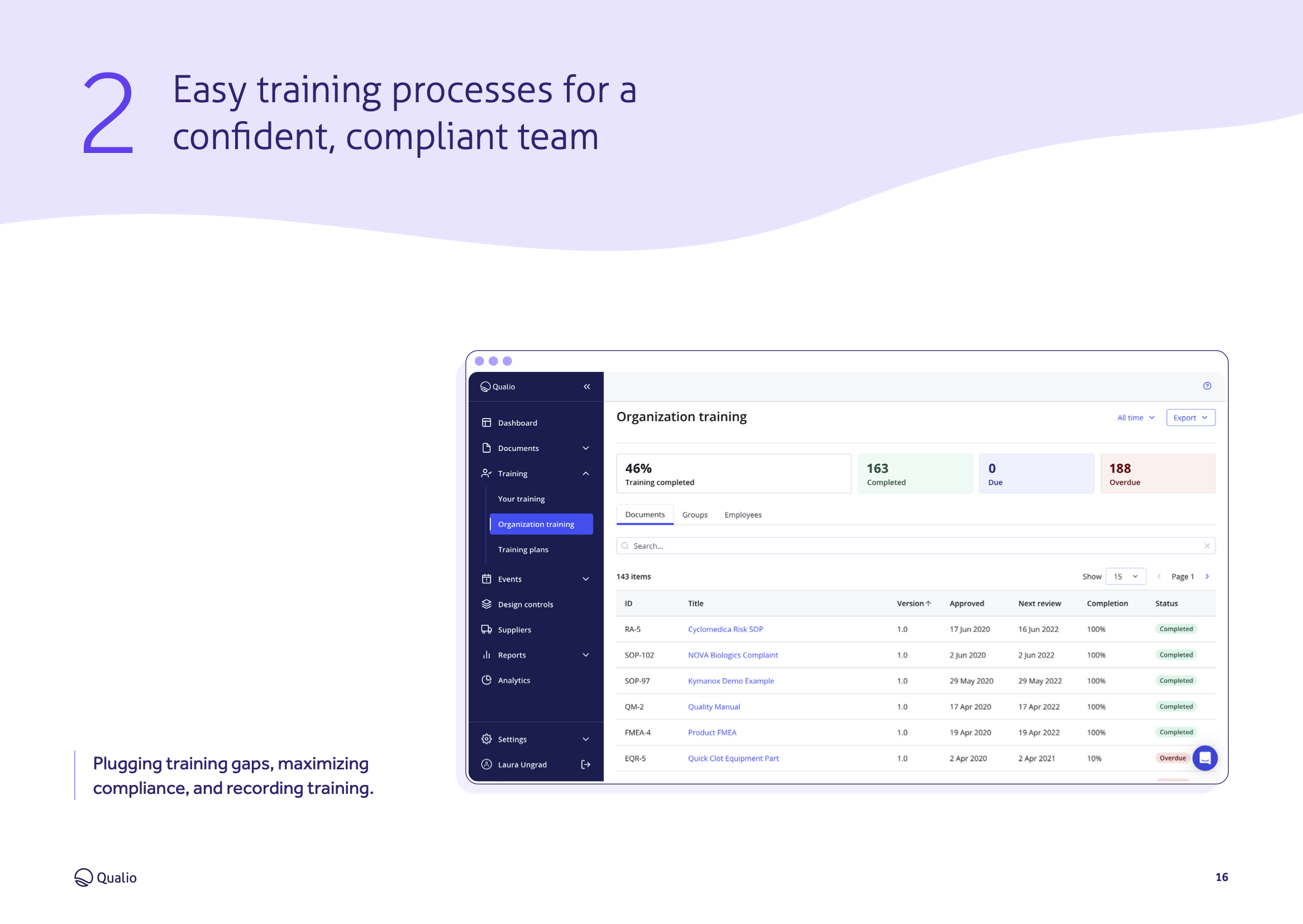
Feature breakdown
Explore the core features and functionality of Qualio, and how each area of the system contributes to a holistic SaMD eQMS
Total compliance
Learn how Qualio meets the demands of ISO 13485, IEC 62304 and more
Best-in-class SaMD quality
Hear from real Qualio SaMD customers, and explore the functionality they use every day