How to prepare for ICH Q9 compliance PDF guide
Master your pharmaceutical or biotech operational risks and put quality front and center.
Learn everything you need to get ICH Q9 compliance in place.
Download our ICH Q9 guide to:
- Understand the core requirements of ICH Q9, the pharmaceutical quality risk management standard
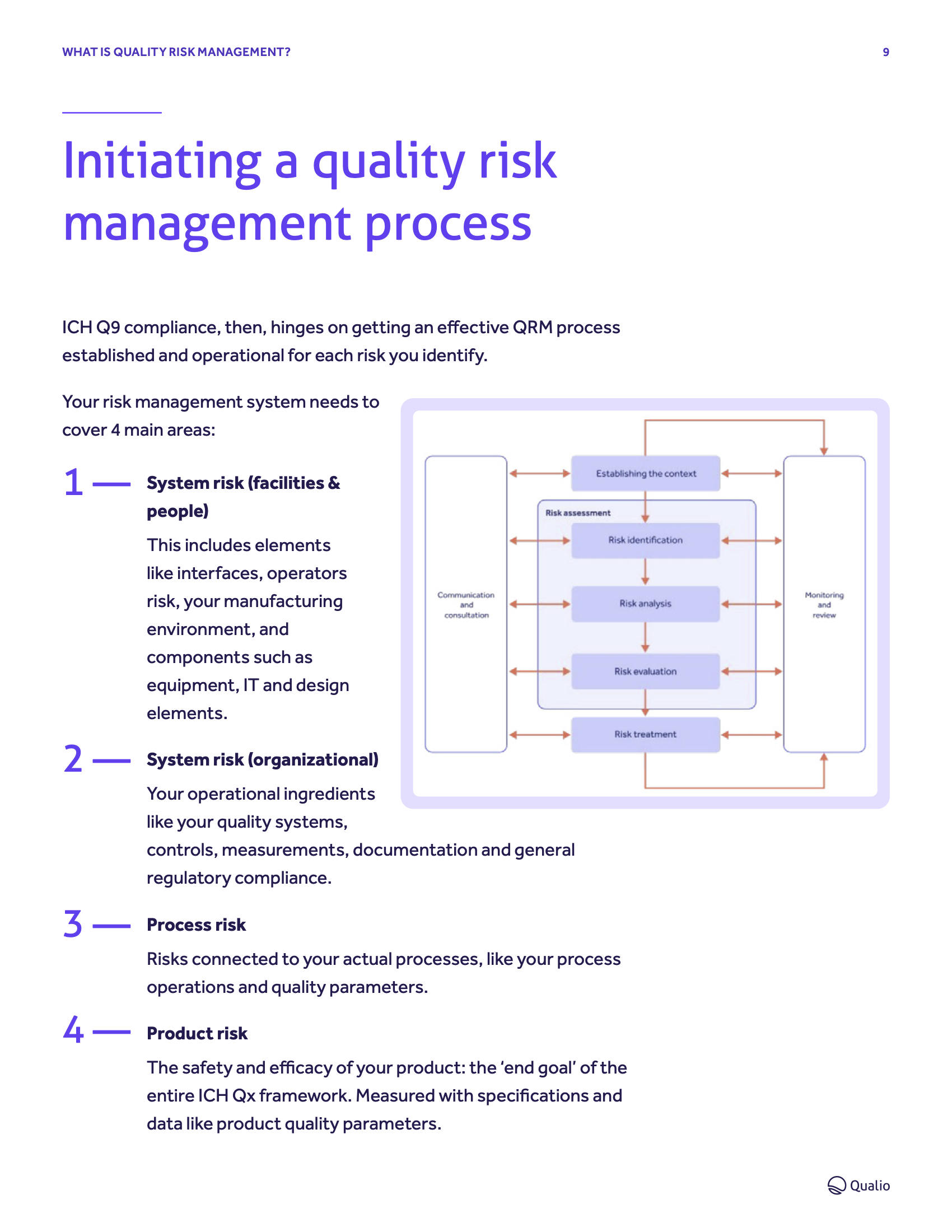
- Master your risks, from identification to treatment, in accordance with ICH Q9 best practice
- Make your pharma or biotech operation safer, stronger and more controlled with proactive risk management
What you'll get:
ICH Q9 compliance guide
Understand what ICH Q9 is and what you need to do for compliance
Pharma risk control
Understand how to practically apply ICH Q9 best practice to control and manage your operational risks
Tips and hints
Understand how ICH Q9 compliance strengthens your pharma or biotech business, know which risk tools to apply, and learn how to integrate quality and risk management for an optimized operation