Bounce back from your PMA submission setback
You're in the right place to turn things around.
Access helpful resources and tools here! 👇
1000+ successful regulatory submissions from our eQMS customers - and counting

Why Class III device companies are tackling their PMA submissions
with dedicated quality software
Get to market and stay there with a powerful eQMS that connects your business
Get the control you need
System guardrails, full digital visibility and expert-built content and workflows get you mature, audit-ready, fully compliant, and ready for a successful regulatory submission
Get to market 300% faster
Centralizing quality and regulatory information in a single source of truth helps you sidestep mistakes and delays, shaving months and years from your market prep process
Get your regulators on side
Close quality gaps, share information with ease and demonstrate airtight digital compliance to unlock market access, secure regulator confidence, and enjoy fewer and faster audits.
Everything you need for PMA clearance
- Align and standardize your product and QMS information with cloud-powered document management
- Build a competent, compliant workforce with digital training
- Fix your submission errors with flexible, collaborative process workflows
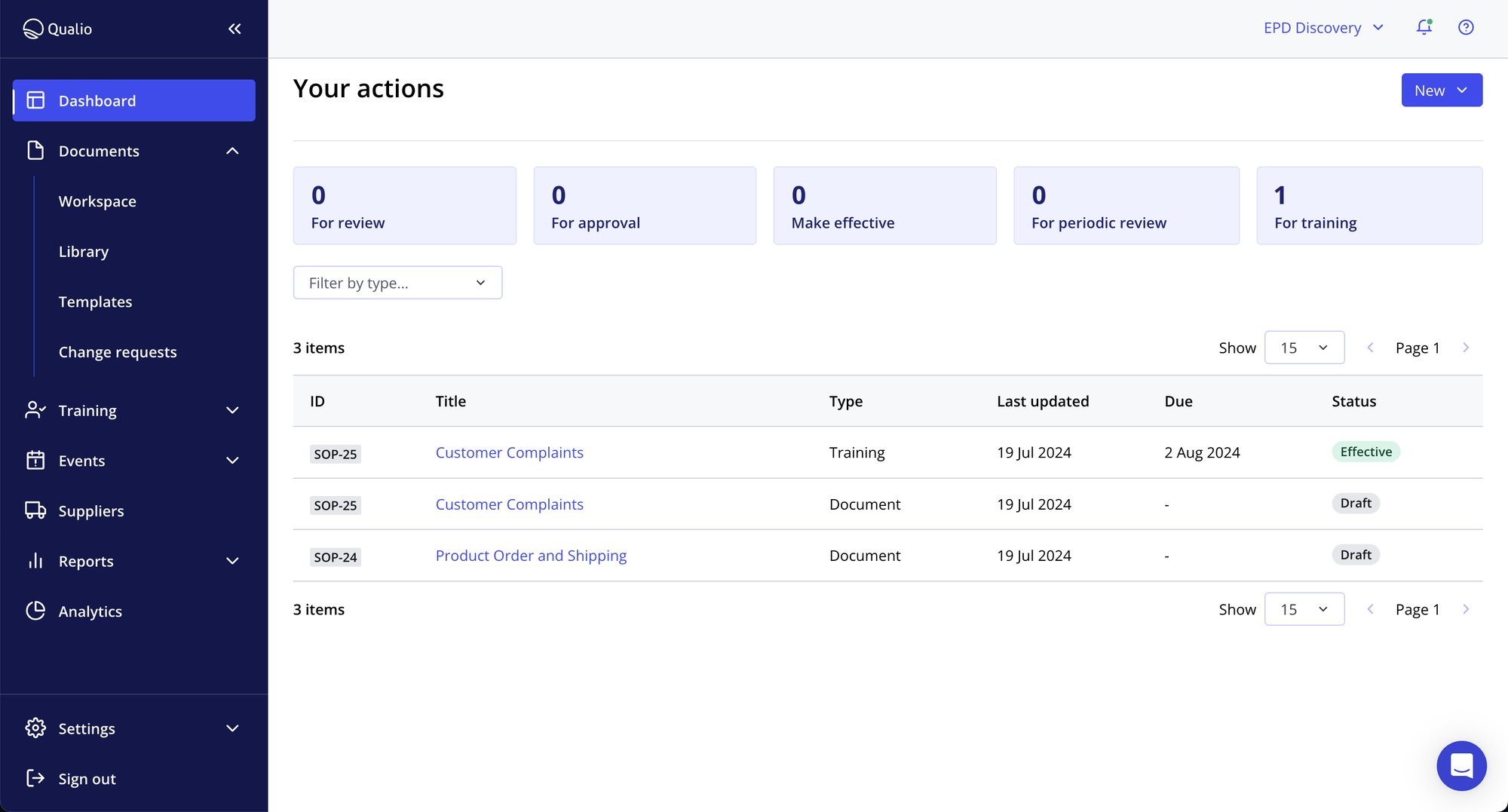
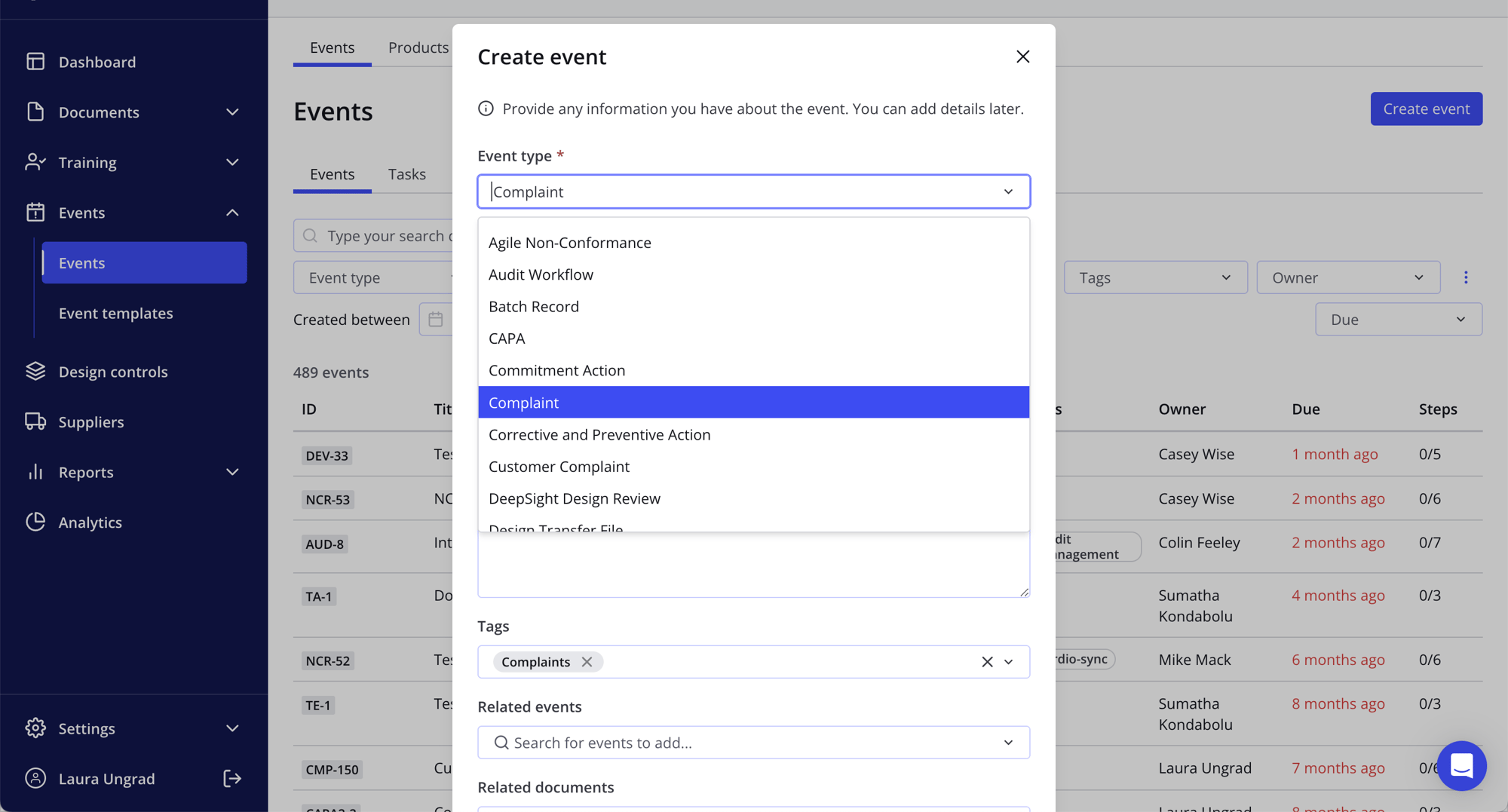
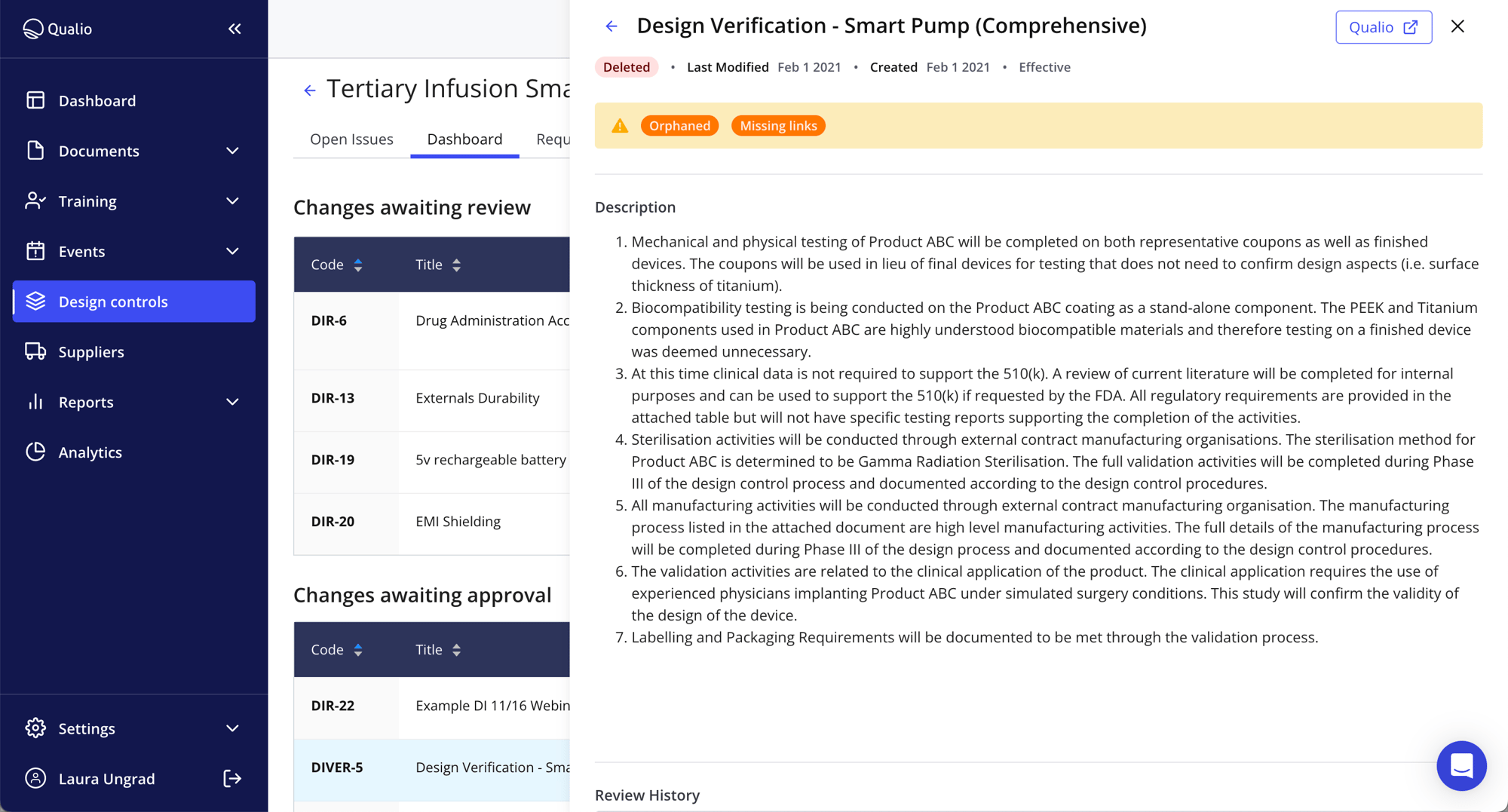
- Grab total control of your medical device risks with fully digitized design control processes
- Optimize your supply chain with holistic supplier management functionality
- Unlock continuous improvement insights with powerful analytic dashboards
Helpful PMA submission resources
PMA checklist
Ace your premarket approval submission with our comprehensive checklist
Qualio datasheet
Explore how our eQMS software arms your company for regulatory success