Your quality management system the way you want it: easy, fast, trusted.
Enjoy less stress and more success getting to market faster with the all-in-one digital QMS trusted by life science professionals around the world.
Talk to an expert today
You're in good company.
Trusted by brilliant life science professionals in over 80 countries.

Quality software + experts on your side.
More success and less stress with everything centralized in one off-the-shelf QMS.
Quality assurance experts on your team.
We provide expertise and clarity on quality best practices so you can tackle challenges in quality assurance, regulatory compliance, audit support, staff training, and more.
We convert your paper-based QMS to digital for free.
We've converted hundreds of paper-based systems to our eQMS and love seeing our customers get hours back each week with automated quality processes.
Ready-to-go content packages for you.
Get procedures, forms, and templates that will save you lots of effort and time during audits and inspections. Training access will make sure that your team is armed with best practices in quality management.
What you'll see in the demo...
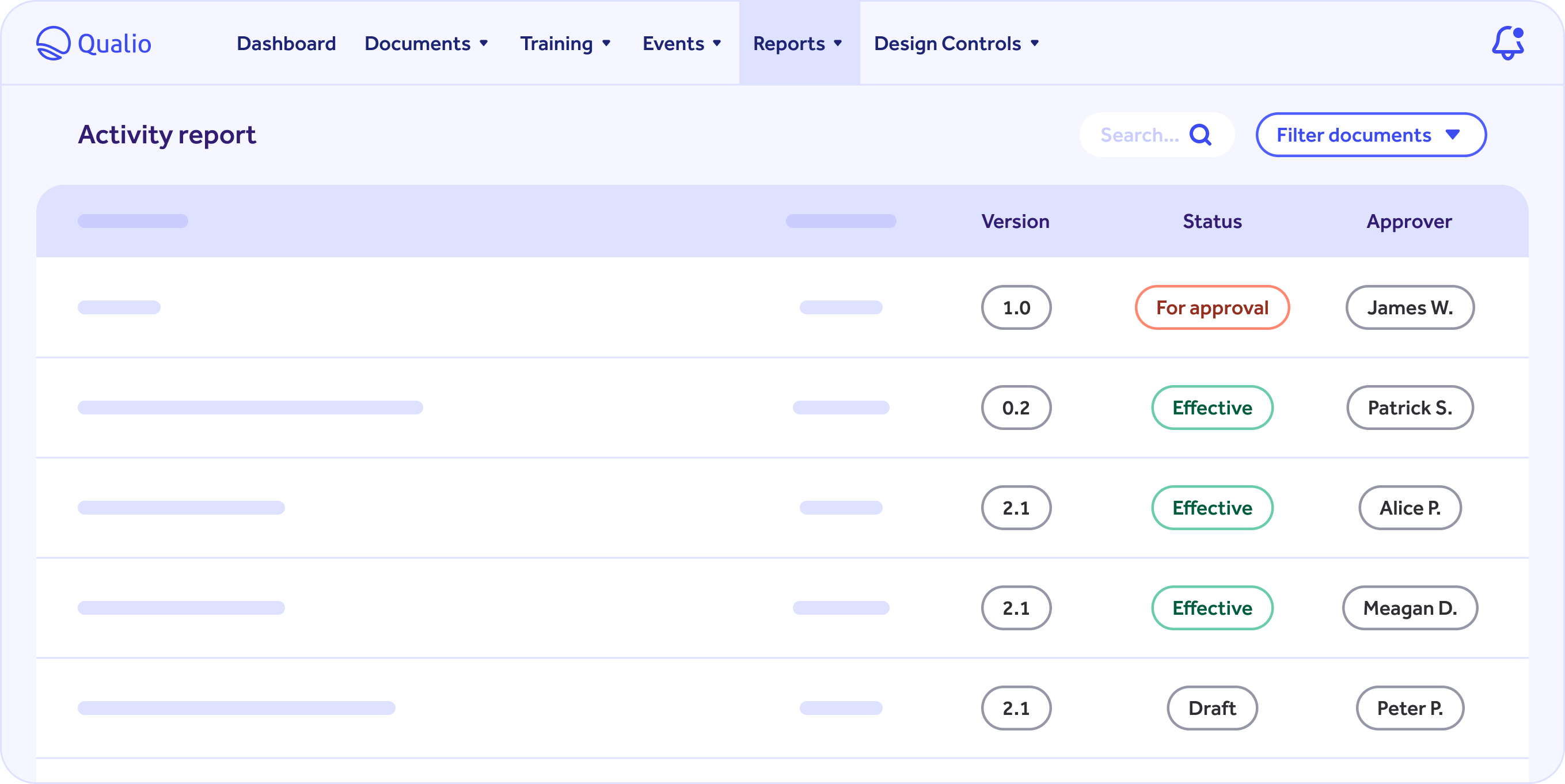
Document Management
- Create new documents from existing templates or upload your existing SOPs.
- Easily find the documents required for audits and inspections with document tags.
- Build workflows for document approval, training, and review period.
Training Management
-
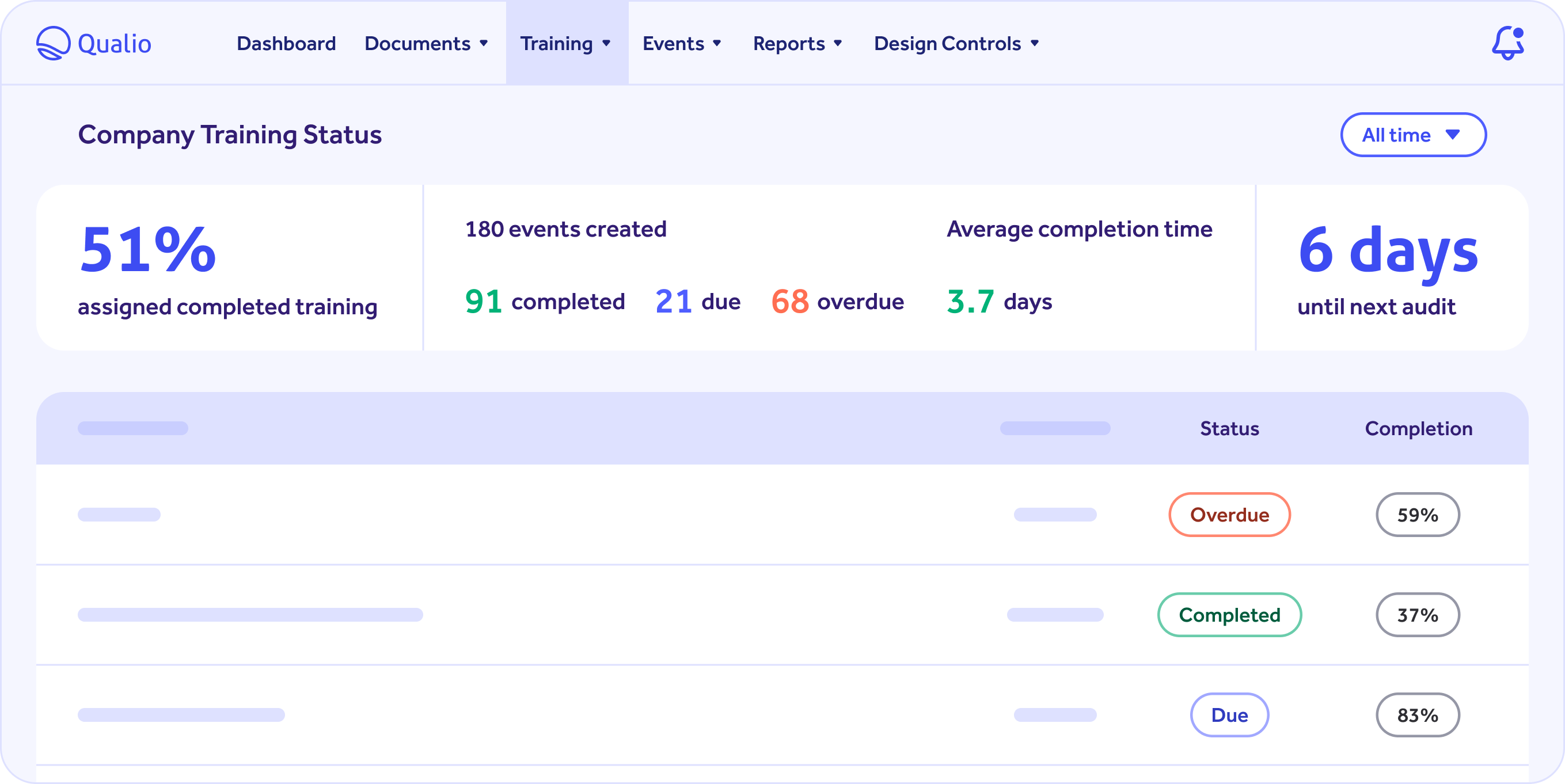
Capture your team's competency with training dashboard.
-
Simple integrated, role-specific training functionality supports in-line assignments as well as tracking of all training activity.
- Save hours by easily sending training reminders to groups or individuals.
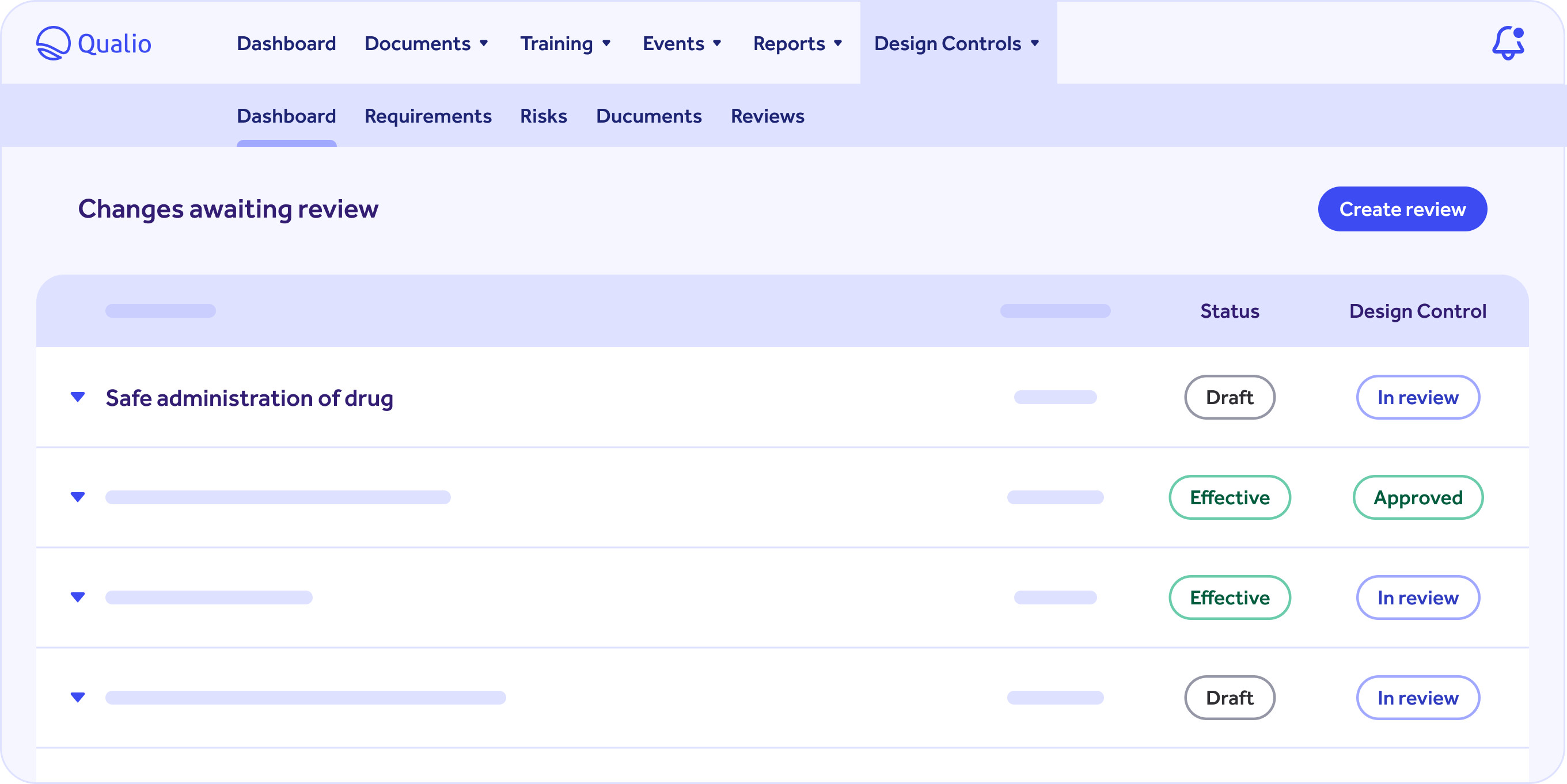
Design Controls
-
Easily meet ISO 13485, ISO 14971 and FDA CFR 820 requirements.
-
Centralize your product data and export key documentation in one click.
-
Focus on building great products with integrations with product tools like JIRA without a compliance headache.
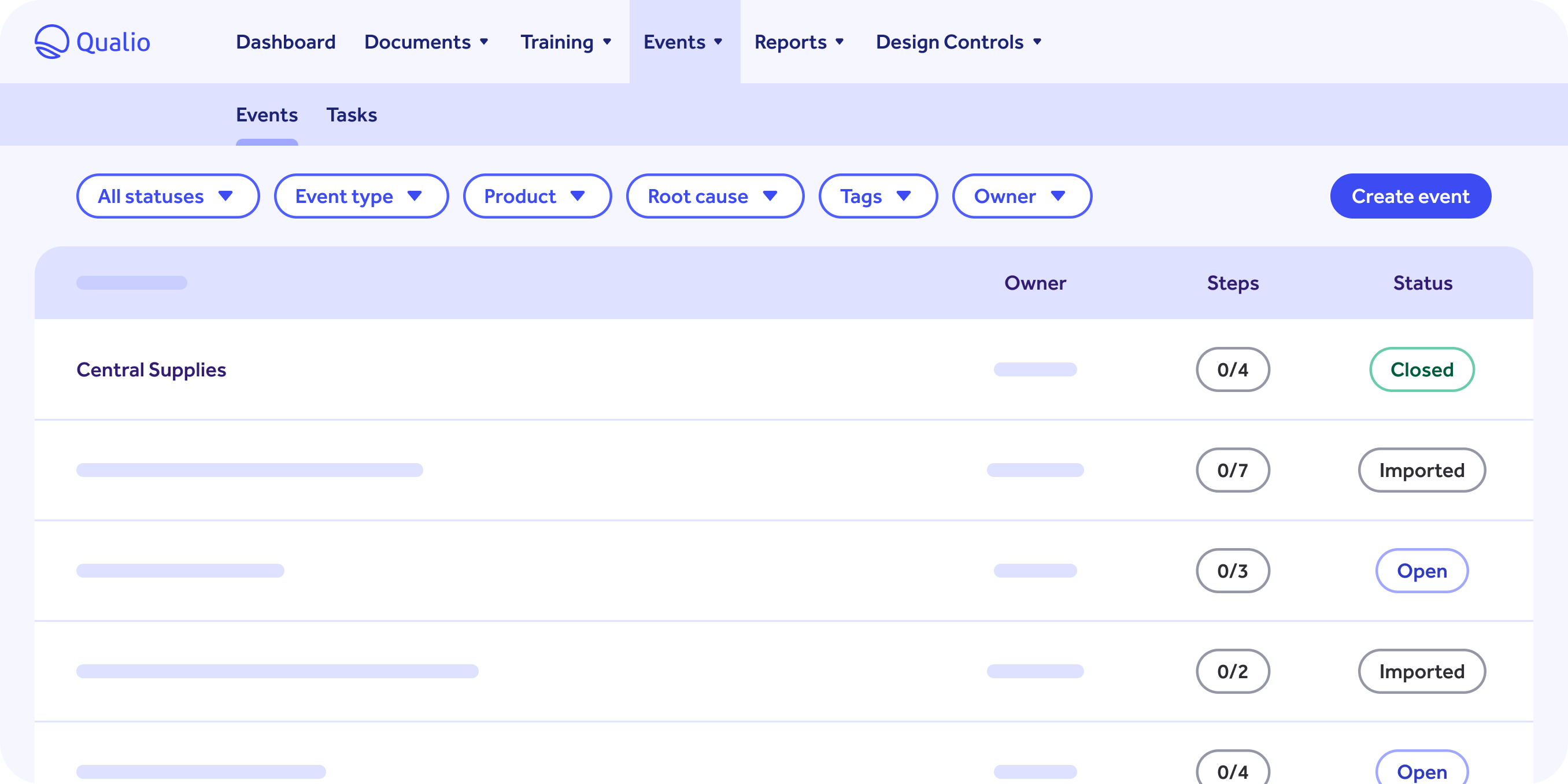
Quality Events
-
End-to-end tracking for non-conformances, unapproved deviations, and customer complaints.
-
See events from creation to investigation, to root-cause analysis, to CAPA.
-
Simple visibility for events through to validation of a successful resolution and event closure.
Audit Ready Reporting
-
Easily see a single place to manage all audits, suppliers, and change management.
-
Be quality driven with visibility across the entire organization in real-time.