CHANGE CONTROL
Change control connected to your entire quality system
Drive effective change control that doesn't slow your business down. Every change is tracked, reviewed, and connected to the documents, training, and CAPAs it affects.

Change management software for your evolving business
Challenge
It can be difficult in a manual paper-based quality system to know the downstream impacts of any change.
Weak change control management can halt your product development process and bring unforeseen risks, complications and defects.
Solution
Qualio’s change management tool gives you total command, control and visibility of any change made in your QMS.
Drill into change histories and audit trails, and drive configurable workflows to manage change properly.
A single change management platform for best-in-class compliance
- Document, review and approve changes on documents, design controls, quality artifacts and more — all from a single change management software system
- Replace insecure and time-heavy paper and spreadsheet processes with automatic change control and recording
- Show changes driven by a quality event, like a complaint or audit finding, to your auditors to demonstrate continuous improvement
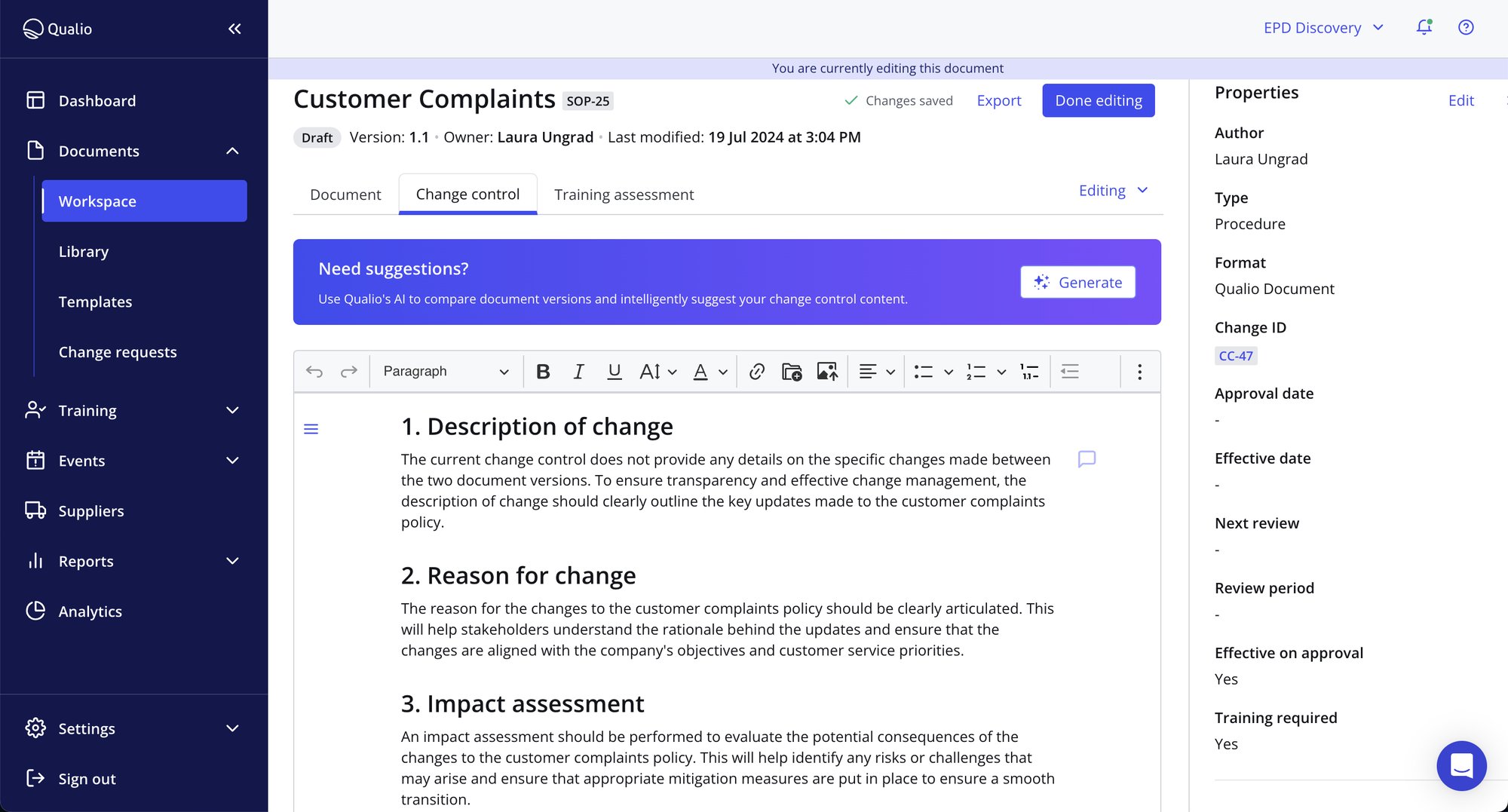
Full traceability and auditability for every change
- Whether a change was driven by an event or an expected process improvement, show the what, when, why, and how of any change
- Ensure everyone is up-to-date on changes with automated notifications, approvals and training workflows
- Connect your team’s tech stack via API integrations, then run change control on artifacts from those systems directly in our change management software
Collaborate on change control management together
- See linked documents as you make changes, set change approvers, manage updates in bulk, and more
- Export change documents and change logs as required
- Stay compliant and audit-ready 100% of the time

Change management software resources

Change management software datasheet
See how our change management software arms your business with flexible action workflows to control any kind of change
Ultimate guide to
change control
Robust change control sets the great companies apart from the rest. Brush up on everything you should know.

What to know about document control software
How can you find the best tool for managing document change control? Find out with our breakdown blog post.
9 core elements of a quality management system
Learn how change control fits within the modern quality management system by exploring the 9 core elements you should get in place.
See other features that make Qualio
the #1 top-rated change management software system
Document management
Make paper, filing cabinets, folders and spreadsheets a thing of the past.
Design control management
Supercharge your product development from a single source of truth.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Supplier management
Control and categorize suppliers and third parties with complete visibility.
Audit management
Make compliance and audit readiness the natural core of your business DNA.