CAPA MANAGEMENT
From detection to close-out in one connected system
CAPA and non-conformance management from detection to root cause to corrective action. Every step tracked, every outcome linked to your quality system.

Why adopt CAPA software?
Spotting and fixing problems is the key to proactive, improvement-based quality management.
But corrective and preventive action (CAPA) and nonconformance management processes that run on outdated legacy tools can no longer keep up with the demands of modern life science.
You need instant visibility of issues, and of who's doing what and when to put things right.
Qualio's CAPA management software gives your business a centralized, workflow-driven platform for driving continuous improvement in a consistent, fully traceable way - without the headache of admin and upkeep.
The #1 CAPA management software for closed-loop quality improvement
Challenge
Your quality teams need established CAPA management solutions for detecting and stamping out defects and weaknesses.
Your auditors expect consistent CAPA and nonconformance management.
And your siloed, paper-based QMS simply can't give you the visibility and control you need.
Solution
Qualio Events operates as both CAPA management software and nonconformance management software, unlocking an efficient, automated pathway for managing all your quality events from root cause to close-out.
Build and apply templates, define steps and tasks, and drive quality improvement that you'll be proud to show your auditor.
Save time with best-in-class CAPA system software
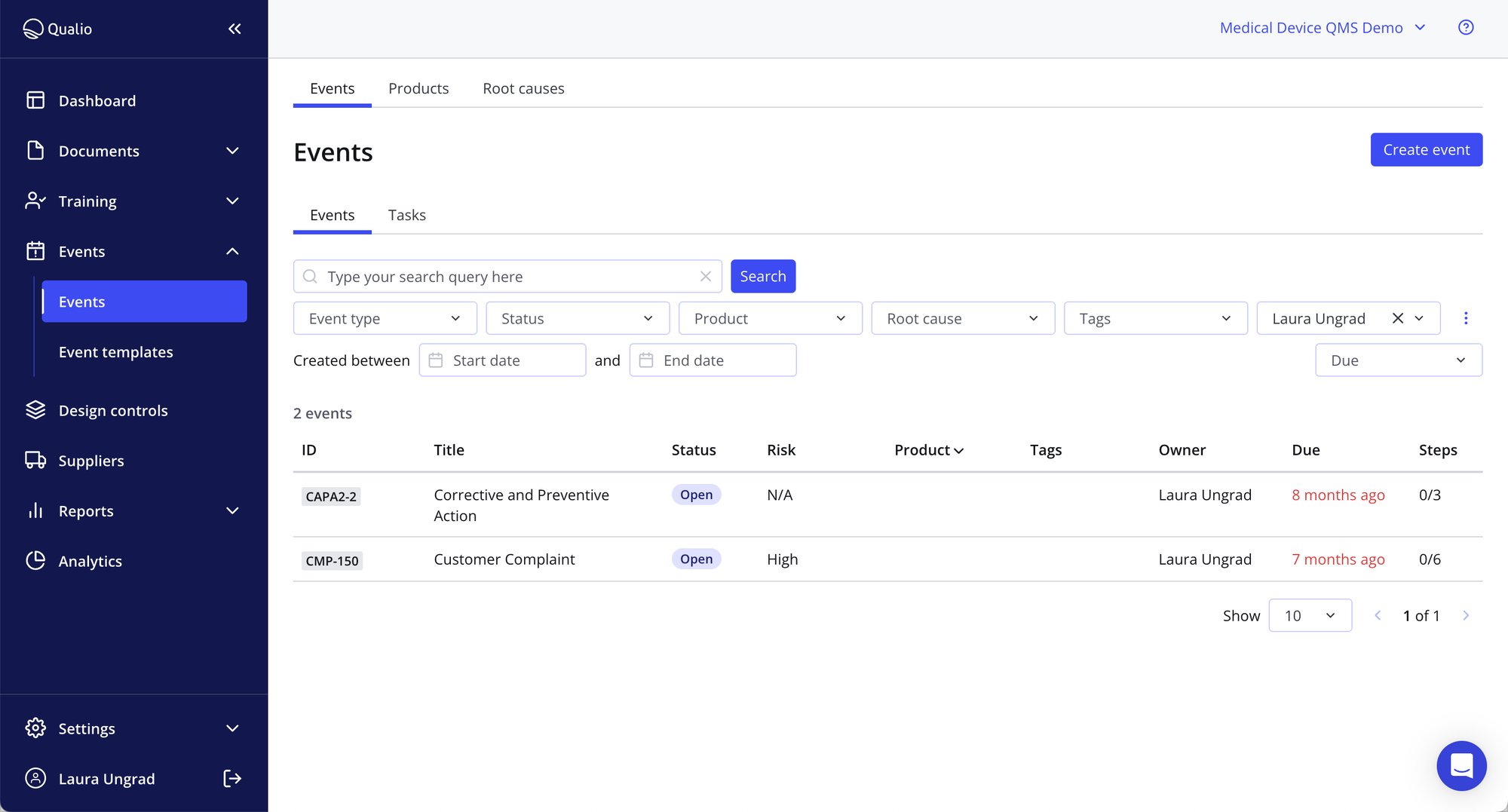
- Build flexible CAPA software workflows with designated action pathways for non-conformances, deviations, incidents, near-misses, complaints and more
- Route tasks, collaborate and comment on events and send automatic reminders to keep your teams aligned
- Stamp out defects and stop them re-occurring with a consistent, traceable approach living in a single non conformance management software area
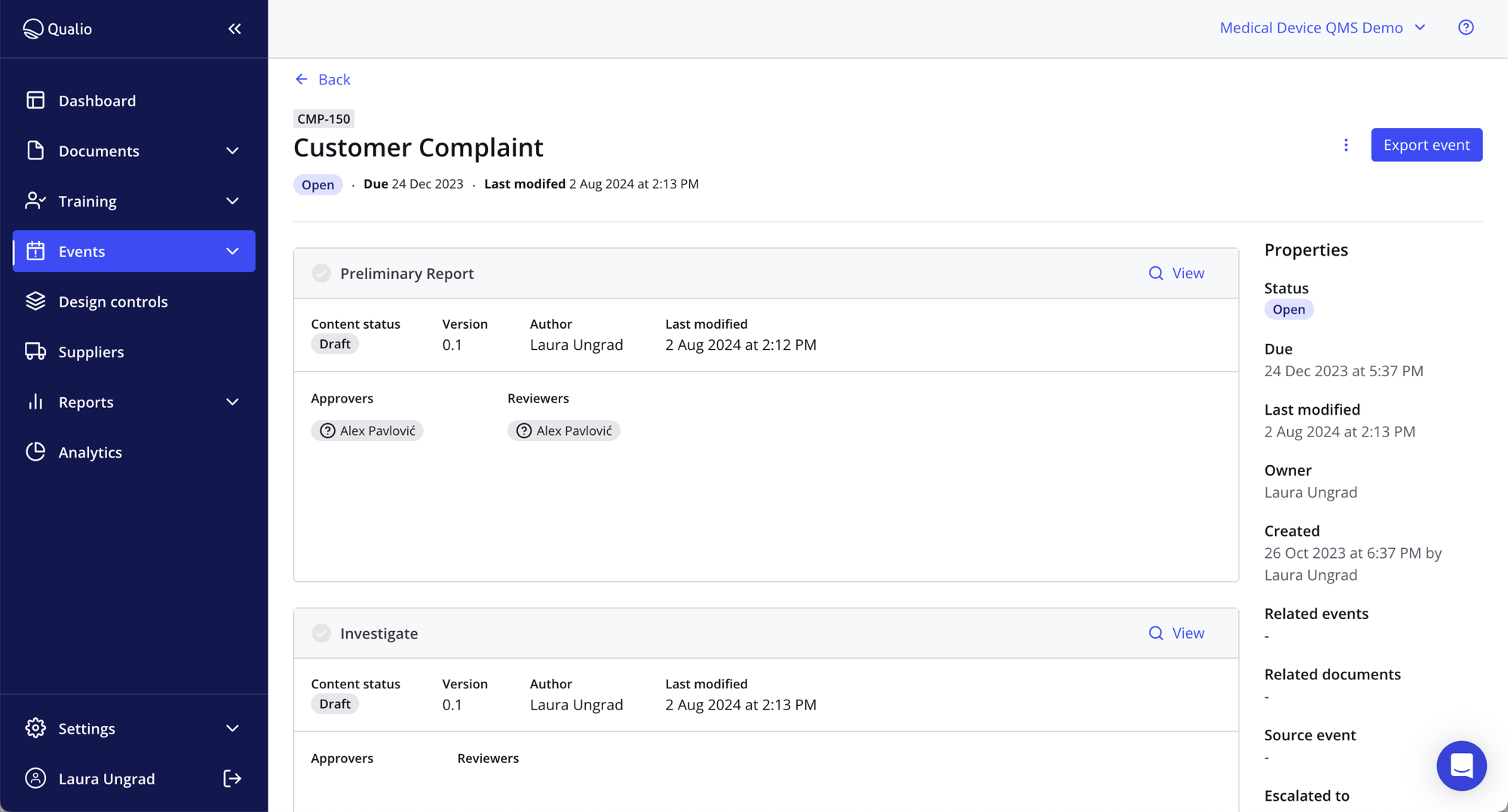
Visibility across your entire organization
- Access powerful reporting on incident rates, closed CAPAs, outstanding tasks and more
- Trace a quality event from beginning to end in a single source of truth
- Connect your CRM, help center and ticketing systems to our CAPA management software to track and manage post-market surveillance and pharmacovigilance activity with ease
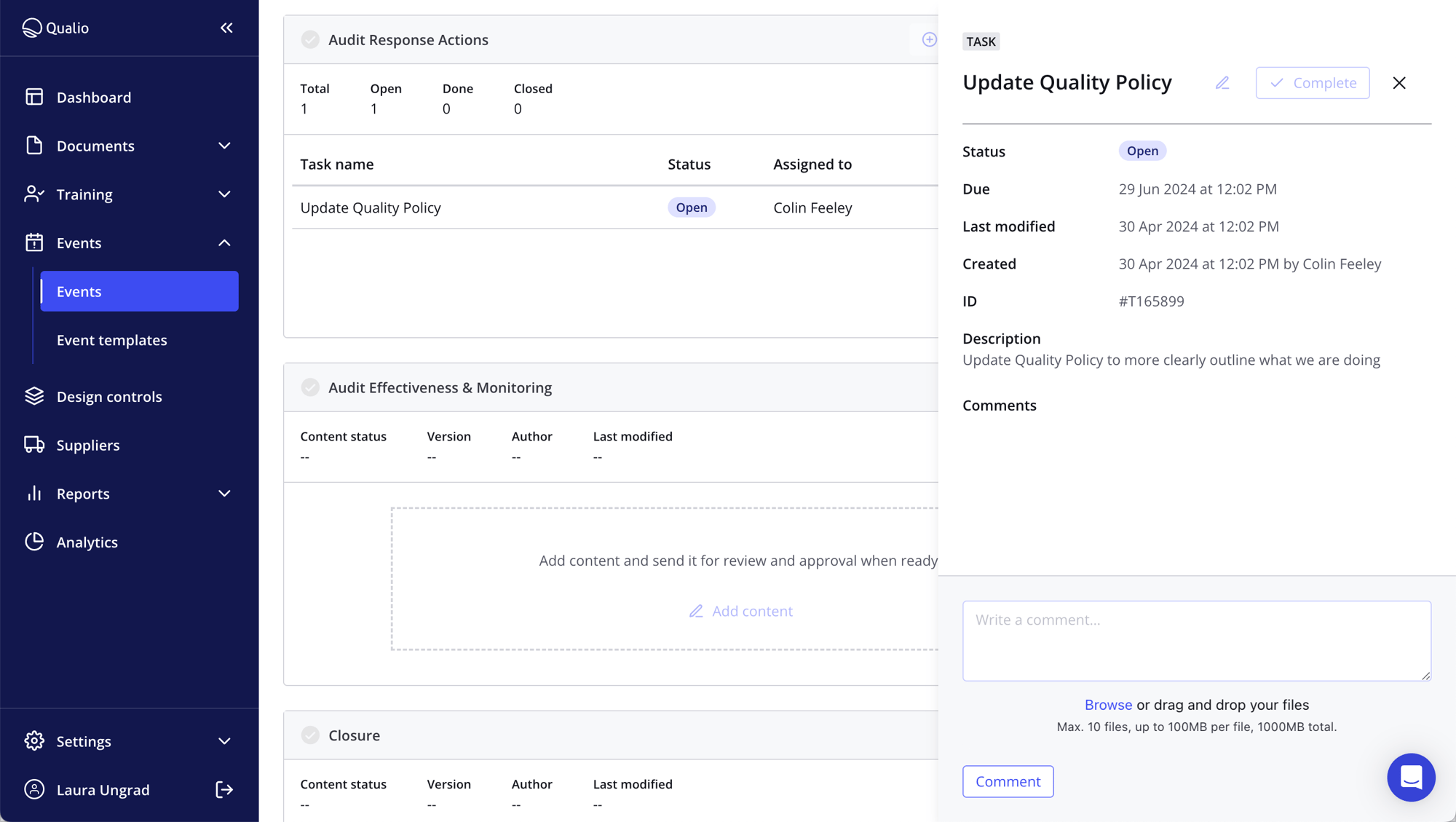
Find the root cause and become proactive
- Find the root cause then fix it, with data at your fingertips for continuous quality improvement
- Cross-reference centralized data objects like customers, tickets and equipment, and link events to documents and training records for a joined-up approach
- Avoid bottlenecks with an automated routing and escalation process to expedite issue resolution
CAPA & NCR management resources

Nonconformance management software datasheet
Learn how our CAPA management software arms your business with end-to-end control of your CAPAs, non-conformances and more

Audit readiness
checklist
Will your CAPA and NCR processes make your auditor happy? Plug gaps and prepare for that next audit with our readiness checklist.
See other features that make Qualio
the #1 top-rated cloud QMS
Document management
Make paper, filing cabinets, folders and spreadsheets a thing of the past.
Supplier management
Design control management
Supercharge your product development from a single source of truth.
Training management
Build a competent, confident, compliant, continuously improving workforce.
Audit management
Make compliance and audit readiness the natural core of your business DNA.
Change control management
Manage change activity easily and effectively with complete traceability.